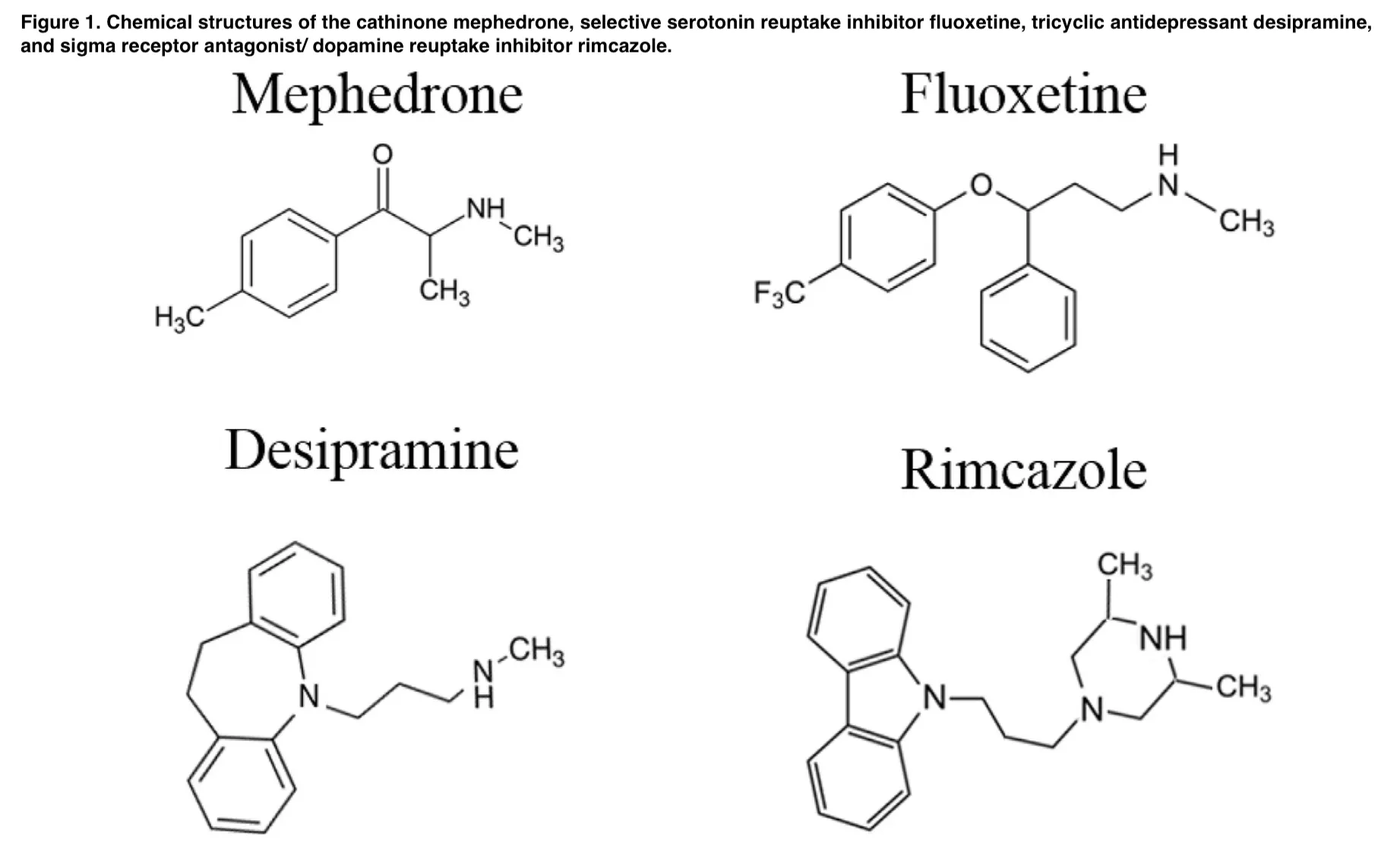
Synthetic or substituted cathinones are known as research chemicals, plant food, glass cleaner or bath salts and labeled not for human use or not tested for hazards or toxicity. Mephedrone 2023 (4-methylmethcathinone) belongs to the class of designer drugs and it is structurally similar to cathinone, which is the principal active constituent of the Khat plant (Catha edulis); also known as natural amphetamine due to its stimulant properties.
Mephedrone shares a number of effects with methamphetamine and MDMA on the central nervous system, such as interaction with monoamine plasma membrane transporters dopamine, serotonin 5-hydroxytryptamine and probably norepinephrine, blocking their reuptake and stimulating their release. Previous studies suggested that mephedrone due to its fact simultaneously on DA release and inhibition of its uptake, could cause methamphetamine-like neurotoxicity and lead to increase the locomotor activity and hyperthermia.
As well as, structural and mechanistic similarities with methamphetamine and MDMA suggests that mephedrone 2023 could also cause toxic effects to dopamine nerve terminals. It has been reported that mephedrone has psychostimulant and hallucinogenic effects, similar to MDMA, methamphetamine and cocaine, has been reported that repeated and regular exposure to mephedrone during the gestational period increases the risk of low birth weight and stillbirth. Also, they reported that repeated administration of mephedrone impairs learning and memory processes through hippocampal damage.
The usage of “bath salt” as reported to poison control centers in the United States has increased at the beginning of 2011. Mephedrone and other synthetic cathinones have been connected to several fatalities in both Europe and the US and are known to have harmful effects on the brain, the heart, and the kidneys.
There is still much to learn about mephedrone’s interoceptive effects and the receptors and transporters that mediate these effects, despite the drug’s long history of abuse recognition. Mephedrone 2023 users have said that MDMA’s subjective effects are the most similar to those of this drug; yet, drug discrimination experiments have revealed an asymmetrical replacement between these two substances.
This study aimed to directly compare and contrast the effects of mephedrone on discriminative stimulus with those of other commonly abused drugs from a variety of pharmacological classes, including mu opioid receptor agonists, DAT inhibitors and substrates, 5-HT2A agonists, and NMDA receptor antagonists.
These drugs, like mephedrone, despite having various mechanisms of action, have been observed to cause a “rush” (heroin), psychomimetic effects (PCP and ketamine), hallucinations (DOI and MDA), stimulant-like effects (cocaine, MDPV, D-amphetamine, methylphenidate). Finally, the contribution of sigma receptors, as well as the NET and SERT, to mephedrone’s discriminative stimulus effects was probed by administering cumulative doses of it after rimcazole, desipramine, and fluoxetine, respectively (Figure 1).
Numerous behavioral effects of DAT inhibitors and substrates, including as cocaine and methamphetamine, are attenuated by rimcazole and other sigma receptor antagonists. A tricyclic antidepressant called desipramine decreases norepinephrine reuptake by binding somewhat selectively to the NET. Desipramine can partially or completely replace cocaine in drug-discrimination tests, similar to mephedrone, and it can also improve the discriminative-stimulus effects of cocaine in rats and monkeys.
The effects of MDMA that are mediated by the SERT and DAT, as well as its subjective and physiological effects in people, have been demonstrated to be attenuated by fluoxetine, which is a member of the family of antidepressants known as selective serotonin reuptake inhibitors.
Methodology of the experiment and materials used
For the experiment, we independently synthesized the racemic form of mephedrone hydrochloride from 4-methylpropriophenone, and the purity of the resulting substance was confirmed by mass spectrometry. Also, in our experiment we used the following substances: cocaine hydrochloride, amphetamine, desipramine, methyl-enedioxypyrovalerone, methylphenidate, and fluoxetine (racemic forms). Rimcazole dihydrochloride was used in the cis-form.
The remaining substances (ketamine, heroin, phencyclidine, methylenedioxyamphetamine and tetrahydrocannabinol) were purchased from various pharmacological representatives. Rats were taught using a fixed-ratio 20 schedule of reinforcement to distinguish between saline and mephedrone. Based on prior studies, the training dose of mephedrone was 3.2 mg/kg. For training, a set daily sequence of injections was employed regularly.
Each session began with a 10-min break, followed by a 30-min FR response phase in which only the assigned lever—mephedrone or saline—was active and generated 45-mg food pellets. During this time, the house light was lighted. Responding to the wrong lever on training days reset the ratio need for the right lever. Following each session, each subject’s reaction rate and the proportion of mephedrone-lever responding were calculated.
Important aspects of mephedrone 2023 research
Initially, rats were deemed to have satisfied the training requirements if they responded to the designated lever more than 90% of the time and the erroneous lever less than 20% of the time prior to the first reinforcement on 9 out of 10 days. When a rat satisfied these requirements successfully, testing with various dosages of mephedrone or other medications got underway, and rats were only needed to meet the training conditions for three days in a row in between tests.
Test sessions included many cycles of the cumulative-dosing process, with the number of cycles representing the number of injections that would take place during that session. Each cycle consisted of a 10-min timeout and a 10-min FR response period.
Depending on the medicine, the delay acted as the pretreatment phase during which subsequent injections raised the cumulative dosage by a quarter. For example, 0.32 mg/kg of mephedrone was injected 10 min before the first test component, 0.24 mg/kg before the second component, 0.44 mg/kg before the third component, and 0.8 mg/kg before the fourth component, thereby producing a cumulative dose-effect curve of 0.32, 0.56, 1, and 1.8 mg/kg for mephedrone.
A larger range was explored in a second session using the same incremental method because this range of doses did not fully capture the dose-effect curve. In order to enhance the number of dosage replications of the training drug, mephedrone, or the other test drugs, overlapping cumulative dose ranges were often evaluated.
During substitution tests only cumulative doses with a single drug were administered. Rimcazole, desipramine, and fluoxetine were tested in conjunction with mephedrone, and a single dosage of each medication was given before cumulative doses of mephedrone. 40 percent propylene glycol, 10 percent ethanol, and 50 percent sterile water made up the rimcazole’s delivery system. 90% saline, 5% emulphor, and 5% ethanol made up the D9THC vehicle. All of the test substances, including mephedrone, were dissolved in distilled or saline water.
Analysis and discussion of the results of the experiment
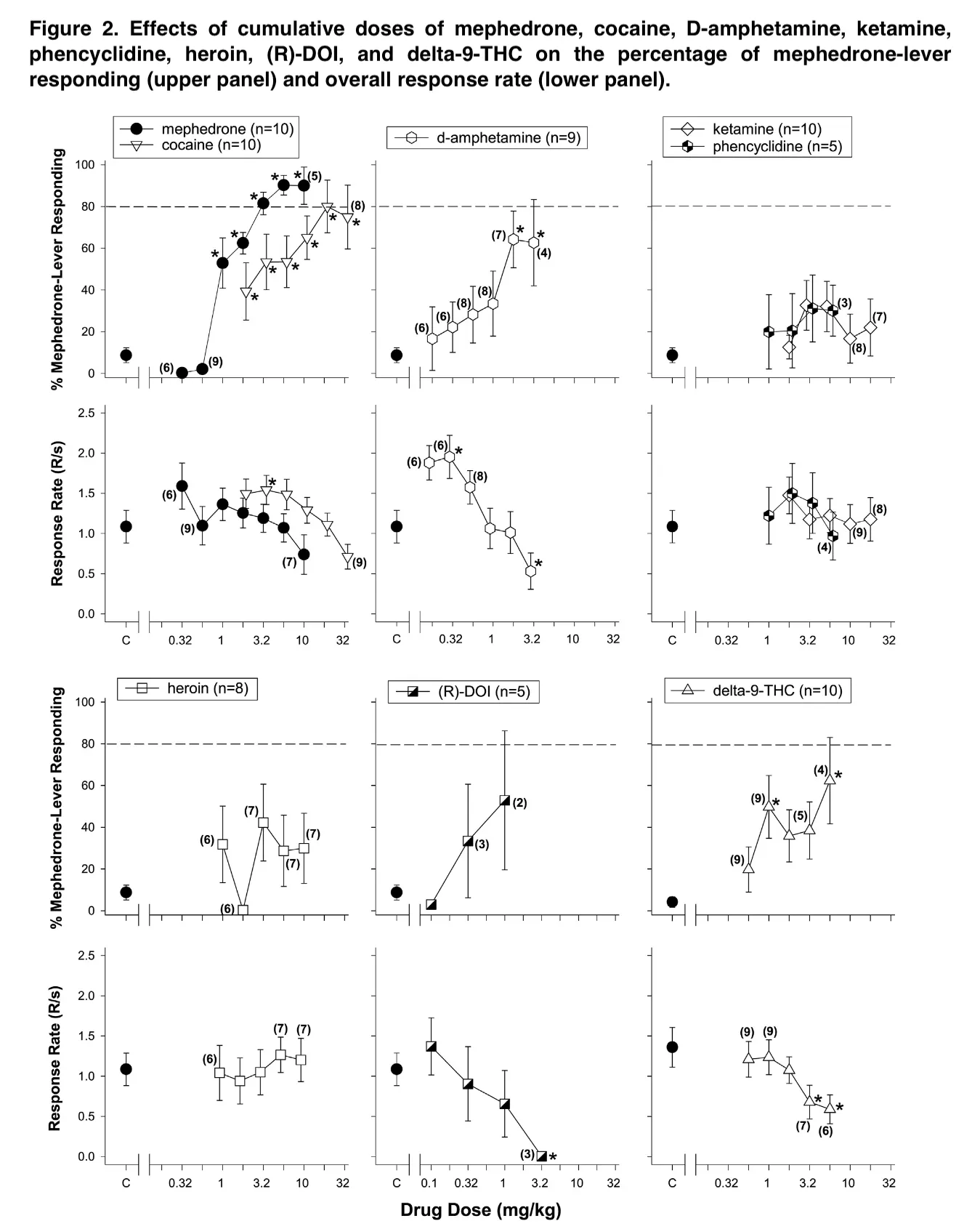
In analyzing the results of our study, we found that the stimulus discriminatory patterns of mephedrone at the dose used were the same, but they were not identical to the effects of some classical psychostimulants. While only cocaine completely replaced mephedrone, other stimulants elicited mephedrone-lever responses of at least 50% before having any discernible effects that slowed down heart rate.
These findings confirm earlier research from our lab showing that methamphetamine and cocaine both dramatically increase the percentage of mephedrone-lever responses without significantly affecting response rates. Earlier studies from this lab demonstrated that, in contrast to these substances, MDMA only induced significant amounts of mephedrone-lever responding at doses that reduced response rate to 26.7% of control.
In support of these substitution data, mephedrone has been shown to substitute in both cocaine- and methamphetamine-trained rats. These findings are also in line with research suggesting that mephedrone may have similar effects to cocaine in terms of inhibiting monoamine reuptake and similar effects to amphetamine in terms of releasing monoamine, both of which are probably mediated through DAT. The fact that mephedrone is cardiotoxic, can be self-administered at levels above those for saline, can elicit conditioned place preferences, induce locomotor activity and sensitize stimulant-induced locomotor activity, disrupt thermoregulation, and increase voluntary wheel-running activity are other similarities between mephedrone and typical stimulant drugs.
Mephedrone’s effects on discriminative stimuli were most comparable to those of medications with greater dopaminergic than serotonergic effects at the training dosage determined for these trials. Despite the perceived similarities between mephedrone and MDMA effects as reported by users, in vivo discrimination experiments have revealed asymmetrical replacement between the two substances. Mephedrone completely replaced MDMA in rats that had been trained to recognize it. MDMA does not completely replace mephedrone in rats that have been trained to recognize it.
The fact that serotonergic medications like DOI and fluoxetine could not substitute for mephedrone but did cause substitution in MDMA-trained rats also points to the involvement of DAT in the effects of discriminative stimuli. Moreover, MDMA could not be replaced by methamphetamine or methylphenidate. This could be due to the differential activity of mephedrone at the three monoamine transporters, NET, DA, and 5HT, and its dose-dependent effects at DAT.
Mephedrone, for instance, has reportedly been found to have higher DAT selectivity than MDMA. In the nucleus accumbens of conscious rats, mephedrone has also been demonstrated to boost dopamine levels more than MDMA and to a higher extent at 3 mg/kg compared to 1 mg/kg. All of these results point to the possibility that the 3.2 mg/kg training dosage enhances dopaminergic effects more than MDMA or lower doses of mephedrone.
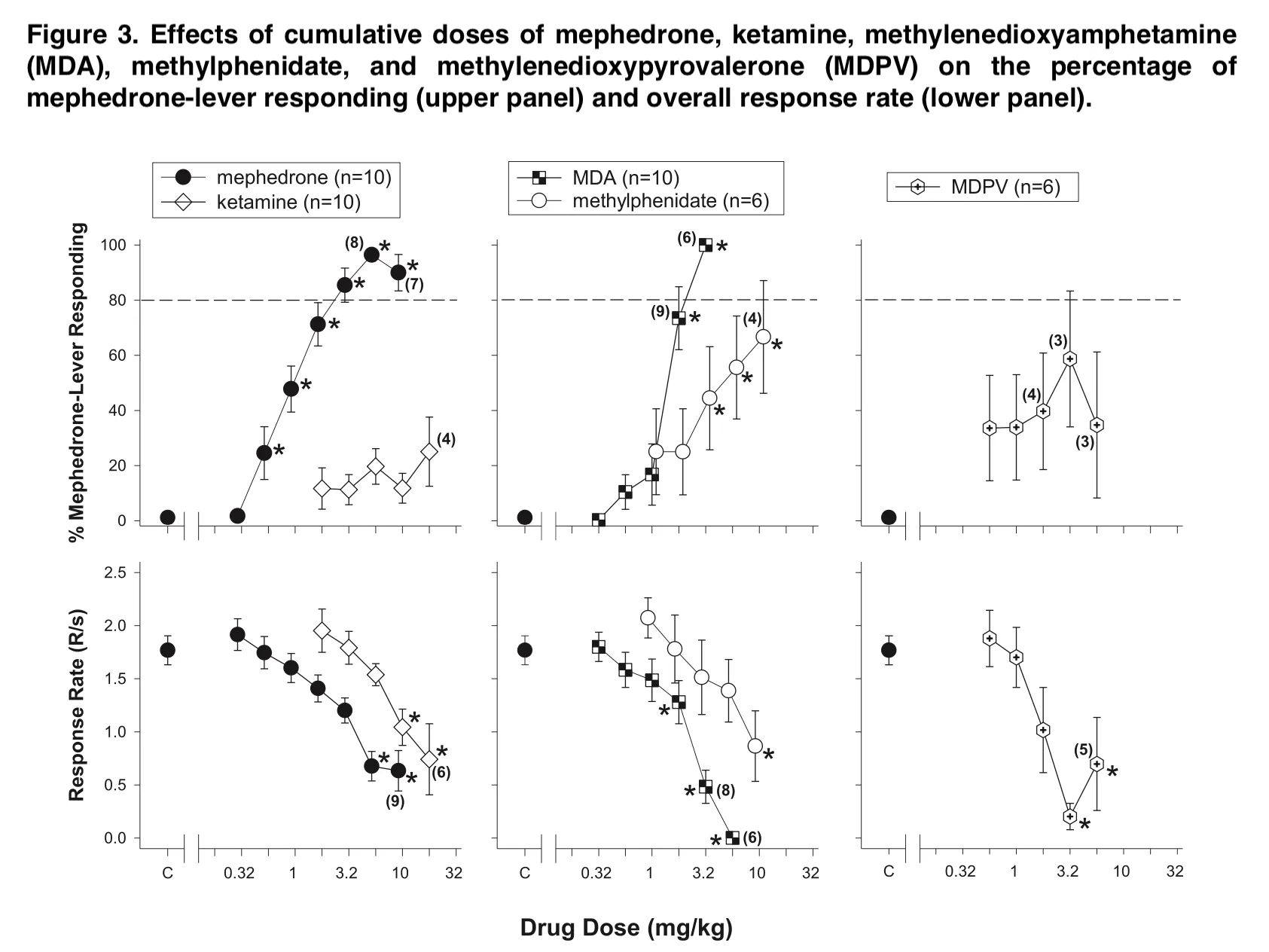
Despite having a structural resemblance to mephedrone, the synthetic cathinone MDPV could only elicit a maximum of 33.89% mephedrone-lever responses at dosages that did not significantly lower reaction rates. Rats taught to distinguish between MDMA or an MDMA/Amphetamine combination and mephedrone also demonstrated differences between mephedrone and MDPV. In contrast to MDPV, which was only substituted in the MDMA/Amphetamine-trained group at levels that also reduced response rate, mephedrone was substituted in both training groups.
This substitution is not particularly surprising and could be explained by data indicating that mephedrone is more serotonergic than MDPV and other pyrovalerone analogs. This may be most evident in monkeys compared to rats where alpha-PVP and methcathinone were found to fully substitute for a cocaine-trained discriminative stimulus, but mephedrone did not produce significant cocaine-like stimulus effects. It is presently uncertain if these variations are caused by a difference in the structural effects of the cathinones in these species, or whether they are a result of species differences.
Mephedrone is both a substrate and an inhibitor of DAT in rats, whereas MDPV exclusively acts as an inhibitor. However, it has been shown that mephedrone substitutes in mice that have been trained to distinguish MDPV, which would indicate another asymmetrical replacement between mephedrone and a medication with a comparable pharmacology.
One surprising result of the substitution testing was that D9-THC partially substituted for mephedrone by creating mephedrone-lever responding more than 50% and considerably boosting mephedrone-lever responding at dosages that did not significantly lower response rate.
Refinement of mephedrone study results 2023
These results could support earlier research showing that D9-THC boosts dopamine release in some brain regions that are also DAT substrates. As previously mentioned, enhanced dopaminergic effects from the 3.2 mg/kg training dosage of mephedrone utilized in this study might further explain why D9-THC can partially replace mephedrone.
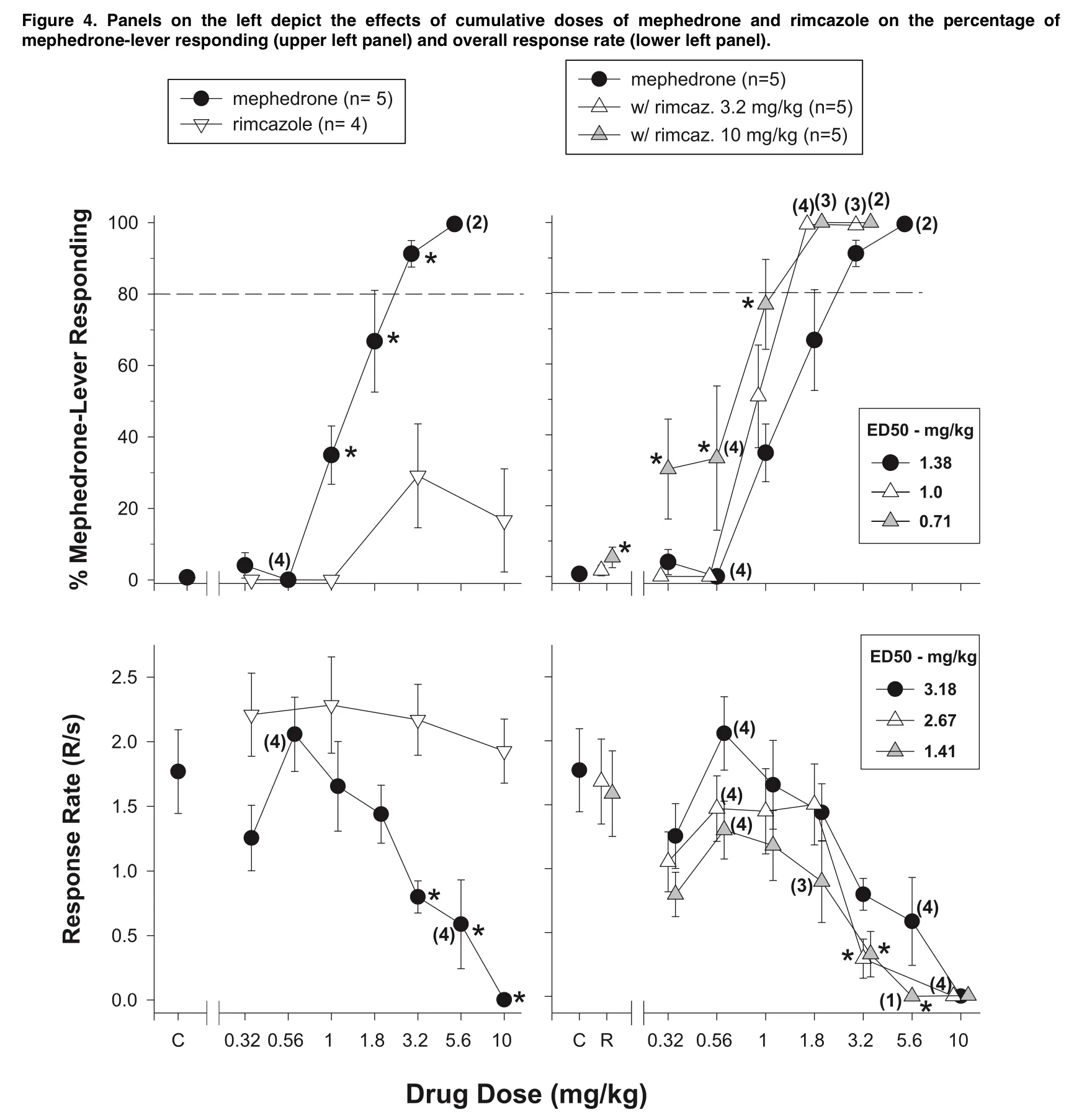
To further understand how the dopamine and cannabis systems interact, more study is required on the role of dopamine in the discriminative stimulus effects of D9-THC. This study also investigated how modifying the activity of sigma receptors and two of the amine transporters might modify the discriminative-stimulus effects of mephedrone in addition to producing data from replacement tests. The sigma receptor antagonist rimcazole was the first pharmacological pretreatment that was investigated.
Rimacazole did not replace cocaine in rats that had been taught to distinguish it, although pretreatment with the medication altered the dose-effect curve for cocaine-lever responding 1.5 fold to the left and the response rate curve downward. Similar to mephedrone, rimcazole potentiated the effects of mephedrone but demonstrated little to no replacement for its effects on discriminative stimuli. Rimcazole lowered the ED50 by roughly two fold and dose-dependently moved the dose-effect curve for mephedrone about one-quarter log unit to the left.
By moving the dose-effect curve southward and lowering the ED50 for the mixture more than two times compared to the ED50 for mephedrone alone, rimcazole also improved the rate-decreasing effects of mephedrone. Rimacazole has a stronger affinity for DAT than sigma receptors, despite the fact that it is classed as a sigma receptor antagonist. Still, rimcazole and other sigma receptor antagonists with affinity for DA have been shown to effectively decrease:
- Cocaine-, methamphetamine-, and MDMA-induced locomotor activity;
- Sensitization to cocaine- and methamphetamine-induced locomotor activity and stereotypies.
- Cocaine self-administration. It has also been demonstrated that sigma receptor antagonists with little or no affinity for the DAT can successfully reduce the locomotor activity caused by stimulants as well as the lethality and convulsions induced by cocaine. However, rimcazole dosages that reduced cocaine’s effects on locomotor activity and reinforcement were much larger than those that increased cocaine’s effects on discriminative stimuli. Therefore, rimcazole‘s affinity for DAT, sigma receptors, or both might have contributed to the potentiation of the discriminative stimulus effects of mephedrone.
Conclusion
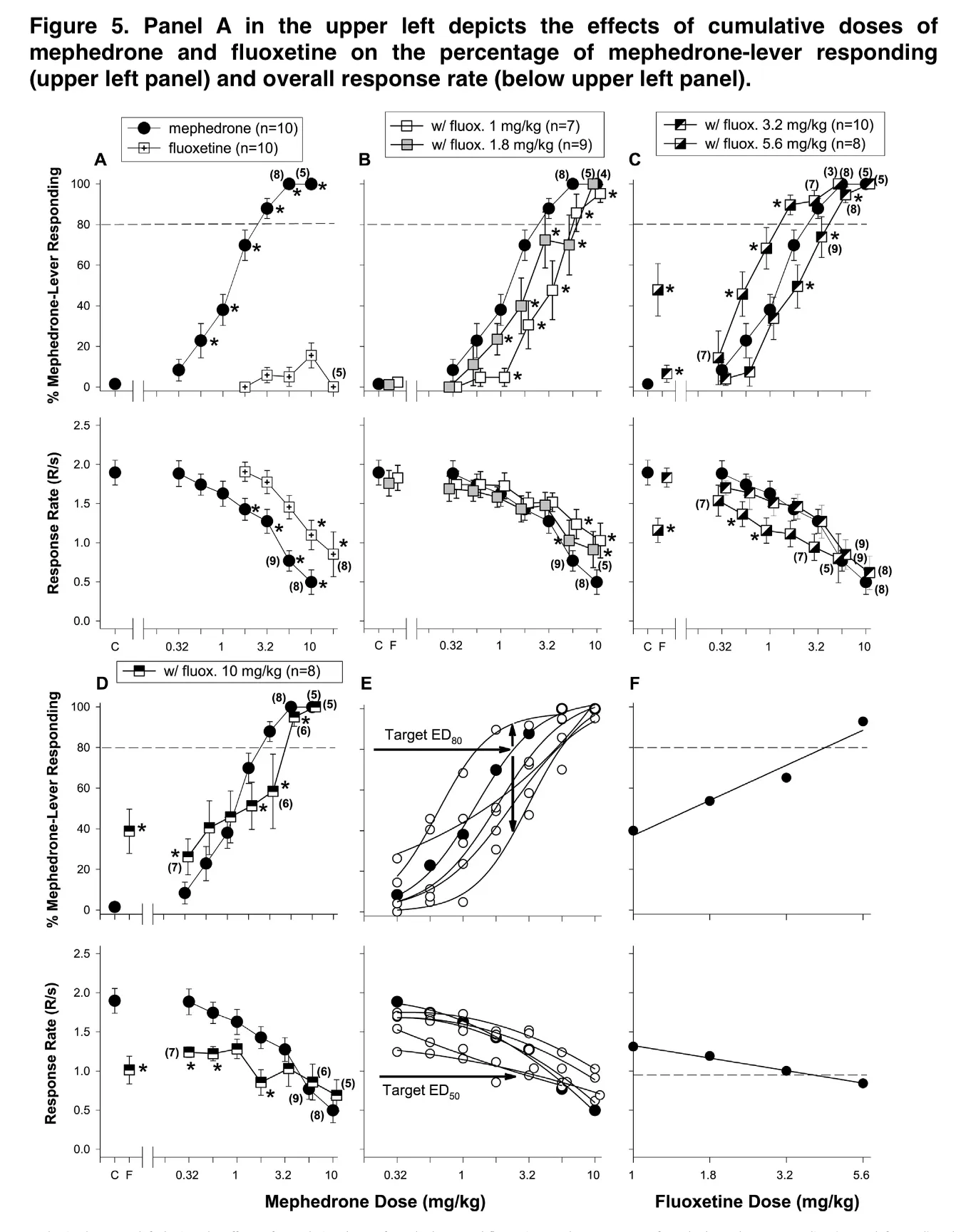
The fluoxetine interaction was the most ordered and dose-dependent of the three interactions evaluated, despite the significant parallels between the discriminative stimulus effects of mephedrone and some typical and non-typical stimulants, as well as its effects at various locations. It was also noteworthy, that the fluoxetine interactions varied from a considerable antagonism at 1 mg/kg to a large potentiation at 5.6 mg/kg.
Furthermore, it is at best challenging to determine whether the effects of fluoxetine on a single transporter or numerous transporters caused these antagonistic interactions. One might assume that the antagonism of mephedrone by low doses of fluoxetine resulted from a competitive interaction between the two drugs at SERT given that the relative in vitro potency for inhibiting the reuptake of serotonin is significantly lower than that for inhibiting the reuptake of norepinephrine and dopamine. Additionally, this would imply that the climbing part of the curve for mephedrone’s effects on discriminative stimuli might represent SERT inhibition, release from SERT, or both.
When considered as a whole, the available findings are consistent with a profile of mephedrone in which its effects on discriminative stimuli are more comparable to those of CNS stimulants, and cocaine in particular, than those of any other class of drugs. The results of the interaction tests further lend credence to the hypothesis that mephedrone functions as a mixed uptake inhibitor and substrate at SERT and DAT, with relatively less activity at NET.
A finding that could only have been made in the present study by testing a wide range of doses in combination with mephedrone is supported by the interaction data involving fluoxetine. This drug can either enhance or antagonize the interoceptive effects of the stimulants, according to Callahan and Cunningham’s observations, which were summarized in their study. The results of mephedrone’s effects on discriminative stimuli may have been quite different if we had simply examined a single dosage of fluoxetine in either the high or low range. The unusual stimulant effects of mephedrone at SERT and DAT will require more investigation.