Mephedrone is one of the most popular synthetic cathinone derivatives used recreationally. Despite being created in 1929, the substance did not garner much attention as a psychoactive agent for a very long time. Mephedrone was launched to the European market around 2007; nevertheless, it was rapidly linked to the emergence of major adverse effects, including fatal intoxications, raising questions about this cathinone’s safety profile. As a result, the chemical was outlawed in the nations that make up the European Union as well as numerous other non-European nations including Israel, Australia, and the USA. However, it is still illegally sold under different street names (e.g., “meow meow”, “bubbles”, “M-CAT”, “cristal bath”, “kati”, “mephisto”) in the form of crystals, powder, capsules, or tablets, and it is used predominantly by oral ingestion and nasal insufflation. However, due to the high solubility of mephedrone powder in water, the drug is also administered via intramuscular or intravenous injection and rectal insertion. It is mostly snorted as a drug. Less often used is intravascular or intramuscular delivery. 93% of its users combine it with other drugs, compared to just 7% who take it alone. Mephedrone has a sympathomimetic effect increasing extracellular levels of aminic neurotransmitters working as a reuptake inhibitor.
Mephedrone exerts its effects by interacting with plasma membrane monoamine trans- porter proteins for dopamine, noradrenaline and serotonin and by increasing the levels of all above-mentioned monoamines in the central nervous system. Mephedrone causes a rapid and dose-dependent increase in both 5-HT and DA levels in the nucleus accumbens (NAc), striatum and frontal cortex in rats. Moreover, it has been shown that repeated mephedrone administration in binge-like regimen causes a rapid decrease in striatal DA and hippocampal 5-HT transporter function in mice which can be perceived as a neurotoxic effect. In addition, mephedrone clearly inhibits DA transporter (DAT) and blocks in vitro DA re-uptake through the VMAT2 in a dose-dependent manner. Thus, an increase in extracellular DA levels following mephedrone exposure likely leads to aberrant DA signaling and stimulant-related behaviors.
Aims and objectives of the publication
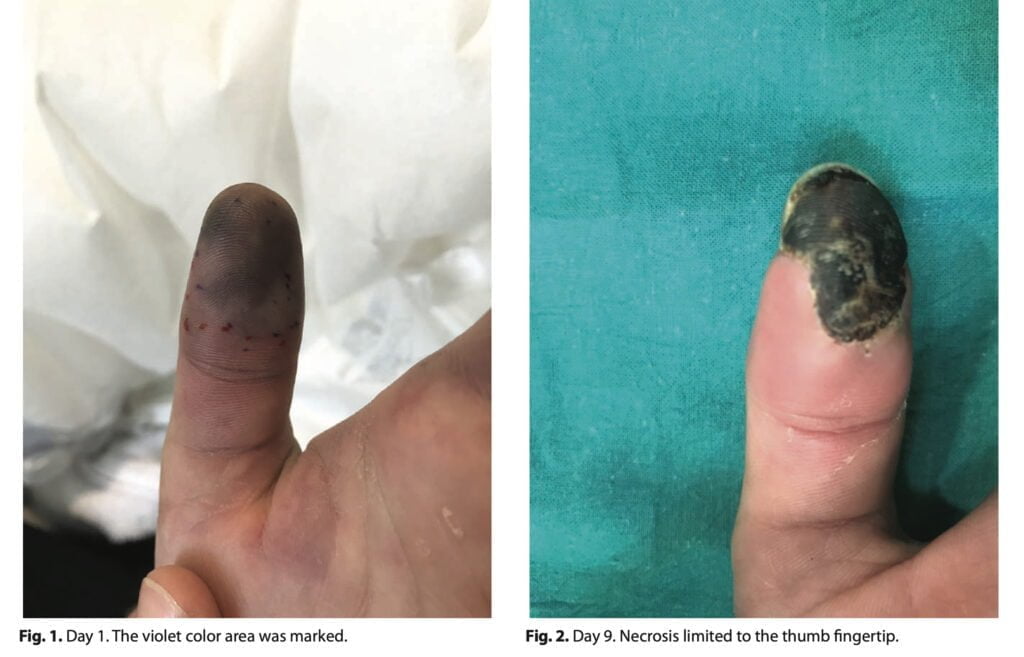
This article’s objective is to demonstrate a clinical instance of distal digital necrosis brought on by intravascular mephedrone injection for recreational use. We conducted a literature search in various scholarly sources using appropriate keys. The subject of our description is a 28-year-old man who has a confirmed diagnosis of HIV infection and is a regular amphetamine user. In October, he went to the emergency room and reported that he had been using large amounts of mephedrone intravenously for the past three days. He had used 4-MMC intranasally for the first few days, then began injecting intravenously for greater effect. A few hours after drug administration, he began to experience edema, a rash, and pain in the left thumb tip. Additionally, he mentioned that he had paraesthesia during the injection in the radial nerve region of his left forearm, distal to the puncture site. The symptoms on his thumb gradually grew worse the next day, eventually becoming violet at the fingertip.
Physical examination of the man revealed swelling of the hand, pallor of the skin on the posterior surface, and purple spots on the distal phalanx of the thumb (Figure 1). The volume of active movements was preserved but limited, the movements were painless, but because of the edema it was impossible to perform full flexion. The remaining fingers had good capillary blood flow. The injection area was located on the volar side of the distal third of the forearm (in the projection of the radial artery). During the clinical blood test, we found pathological parameters. Ultrasonography revealed edema and necrosis in the area of the scapular muscle. There was no chest discomfort or other cardiac symptoms, thus an echocardiogram was not done. In the emergency room, we gave him 10 mg of nifedipine. The patient asked for voluntary discharge against the medical criteria and decided to come back the next day. When he was hospitalized, we noticed that the cyanosis had advanced to the tip of the thumb finger and the radial side of the index finger.
After hospitalization, the patient received the following therapy: 10 mg nifedipine every 8 hours; 1 gram of amoxicillin with clavulanic acid every 8 hours intravenously; 40 mg of enoxaparin subcutaneously every 24 hours; 2 grams of metamizole intravenously every 8 hours and one gram of acetaminophen intravenously every 8 hours. Additional treatment includes refraining from smoking, resting the extremities, raising the left arm, and using a heated blanket (36–40°C) continuously throughout the day. The patient was checked by the infectious disease team while they were keeping them in the hospital to start HIV therapy.
Dynamic observation and the result of therapy
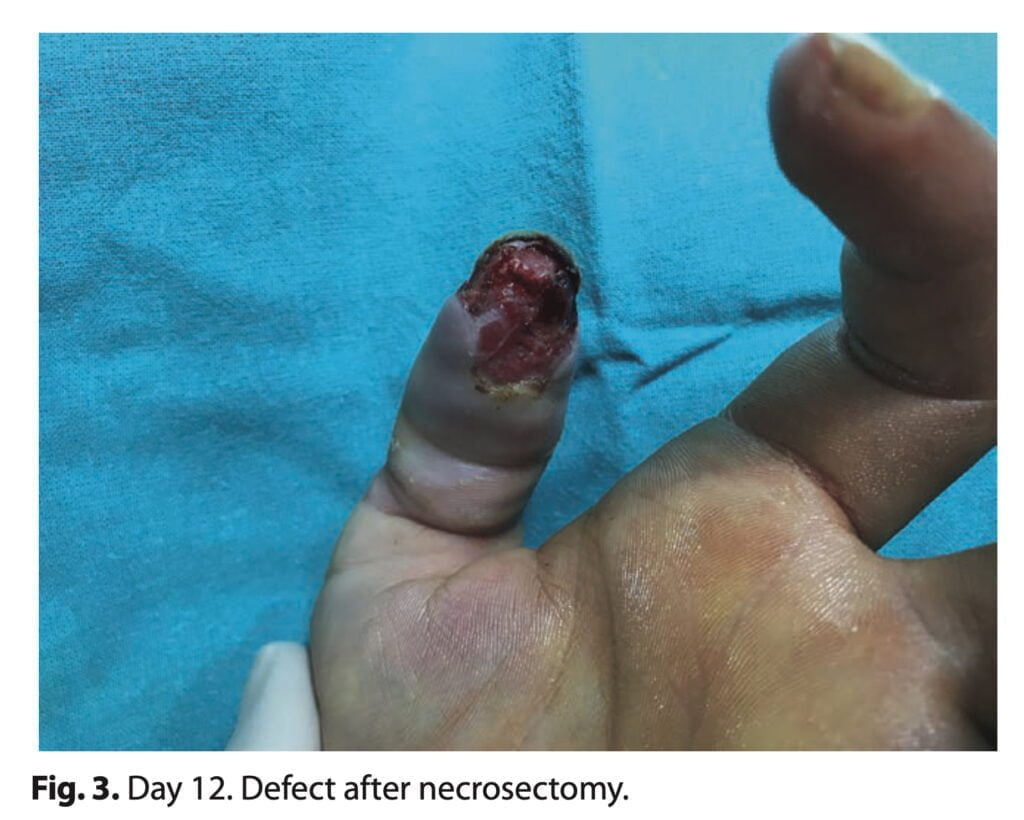
We saw continued cyanosis at the thumb tip during the hospital stay, with complete resolution in the other places. At the injection location in the forearm, there was no skin necrosis visible. Unknown is the cause of the necrosis’s exclusive localization to this region. Day 9 saw the establishment of a well-contained region of necrosis (Figure 2). The x-ray findings showed no indication of bone necrosis at any point. Because of the semiology of the injuries, we supposed that the patient incidentally administrated himself the drug either in the proximity or intra-arterially to his radial artery. Numerous mechanisms, such as vasospasm, medication embolism, thrombosis, or arteritis, might be to blame for the necrosis. It’s also possible that undiagnosed underlying disorders from the past, including connective tissue diseases, played a role.
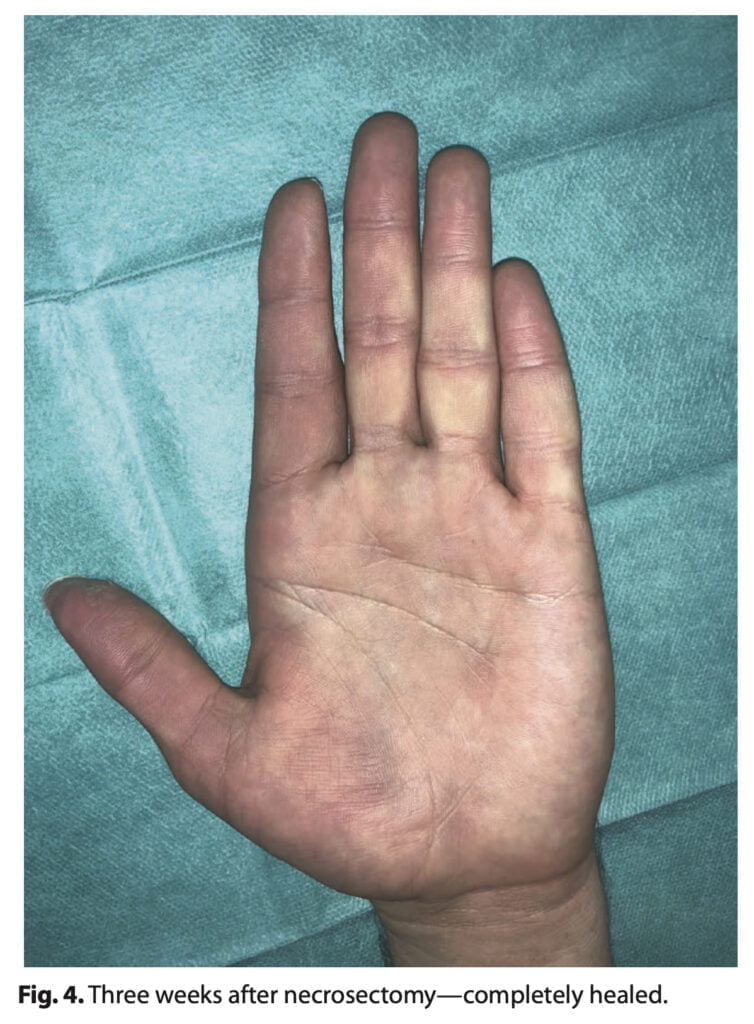
In our clinics, the patient was given conservative care. After performing a superficial necrosectomy, we created a 4 cm-wide gap in the skin and subcutaneous tissue (Figure 3). No exposed bone could be seen. Every other day, Linitul and gel iodized antiseptics were used for wound care. In 3 weeks, the imperfection was fully repaired per secundam, returning the pulp’s sensitivity and function to normal (Figure 4). Surgical management was dismissed because of patient characteristics. A possible shortcoming of our treatment is that healing per secundam does not ensure best sensitivity.
Conclusion

The patient mentions symptoms like secondary Raynaud’s syndrome linked to sudden temperature swings as a single long-term physical injury (Figure 5). This can be explained by sympathetic hyperactivity brought on by the chemical toxicity of mephedrone in the artery wall, which occurs as a side effect of various chemotherapics. If the symptoms persist for an extended period of time, perivascular injection of a calcium channel blocker or local anesthetic as a chemical sympathectomy will be explored. Complications related to changes in local intravascular clotting have not been reported. A potential side effect of mephedrone use may be necrosis of the distal phalanx of the fingers, but the mechanism of this complication is still unclear.
