Mephedrone affects reward from morphine and causes organ damage
Mephedrone and morphine: why is this combination of psychoactive substances the most dangerous? Why are human organs damaged faster? How can organ damage be prevented when 4mmc and morphine are used together? The answers to all these questions, as well as many others, can be found in this publication.
The usage of what are known as “legal highs”, or psychoactive drugs, has significantly increased in recent years. Mephedrone, a synthetic cathinone, is one of them. Low dosages of mephedrone have a psychostimulant-like effect. However, it might cause major side effects including hypertension, tachycardia, aggressive behavior, and other peripheral issues at larger dosages or after prolonged usage. Mephedrone has characteristics with other psychoactive substances like amphetamine and cocaine in terms of its pharmacological action.
Mephedrone’s mechanism of action relies on altering dopamine and serotonin release and turning on their transporters in the brain. Mephedrone misuse, particularly in adolescents, has been linked to long-lasting changes in brain activity, according to research in the literature. More research is being done on the effects of mephedrone that is created throughout adolescence since these mesolimbic system disruptions may last for months after the last drug administration.
Mephedrone and morphine one of the most dangerous combinations of psychoactive substances
For instance, Ciudad-Roberts found that the combination of ethanol and mephedrone disturbed the oxidative stress-related enzymes, causing neurotoxicity and impairing neurogenesis and learning in adolescent mice. Motbey showed changes in the distribution of Fos expression in adolescent rats after mephedrone administration. Mephedrone may potentially affect a person’s peripheral organ function.
Although morphine is an effective painkiller, its use is made challenging by the onset of tolerance and physical dependency. Gamma-aminobutyric acid (GABA) terminals in the ventral tegmental region contain opioid receptors that are stimulated by morphine to provide the rewarding effect. The conditioned place preference (CPP) test has been used several times to evaluate the rewarding effect of morphine.
The rewarding impact of morphine may be altered by a number of variables, including dose, mode of administration, the person’s drug history, food, and stress, which can cause genetic and/or epigenetic changes. One of the epigenetic processes is DNA methylation, which involves the addition of methyl groups to the DNA molecule, primarily at the 5′ position of cytosine nucleotides. DNA methyltransferases modify DNA sequences enzymatically. It could modify gene expression and change how the brain functions. In the current study, we set out to determine if a rat’s exposure to mephedrone as a teen affected how pleasurable it was to receive morphine.
As a result, we ran the CPP test, a behavioral experiment. The degree of striatal DNA methylation was then examined. We also conducted histopathology tests to see how teenage mephedrone exposure affected the function of the kidney and liver in adulthood. Our work is the first to examine the long-term effects of mephedrone on the rewarding activity of morphine in rats, despite the fact that other papers have indicated a variety of mephedrone effects. Our study also aimed to evaluate the influence of adolescent exposure to mephedrone on structure of peripheral tissues.
Methods and materials
In the study, we used male Wistar rats weighing 200-250 grams. The following substances were used in the behavioral experiments: morphine hydrochloride and mephedrone hydrochloride. These substances were dissolved in saline and injected intraperitoneally at a dose of 5 mg/kg for morphine and 10 mg/kg for mephedrone. Three weeks following the last injection of mephedrone, on the 60th PND, the CPP test using a biased procedure was carried out. Eight rectangular boxes made up the applicable equipment; each was divided into three compartments, two of which were segregated from a tiny, gray region in the middle by detachable guillotine doors.
The two big compartments featured different colored and patterned walls and flooring; one had black walls and a white mesh floor, while the other had white walls and a black, solid floor. A digital video camera system mounted immediately above the testing device tracked the subject rat’s location during the experiment, and a video tracking program automatically documented how much time each rat spent in each of the two sizable compartments. The CPP test consisted of three, typical phases: preconditioning, conditioning, postconditioning.
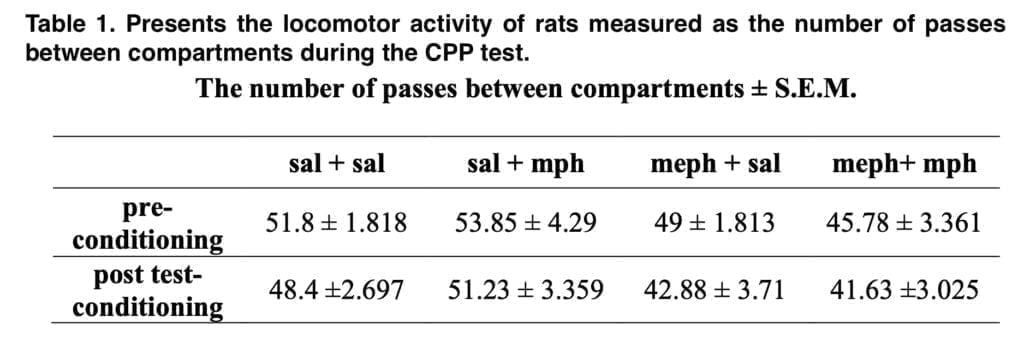
The locomotor activity of each rat was counted during the 15 min CPP test as the number of crossings between compartments. The rats were decapitated after the behavioral tests were completed in order to dissect the striatum for epigenetic research. The animals’ kidneys and livers were also removed for histological analysis. DNA was isolated from the striatum according to the included protocol. Total DNA methylation was determined according to the enclosed instructions using a kit for quantitative analysis of DNA methylation. The procedure consisted of three stages: DNA binding, methylated DNA capture and detection.
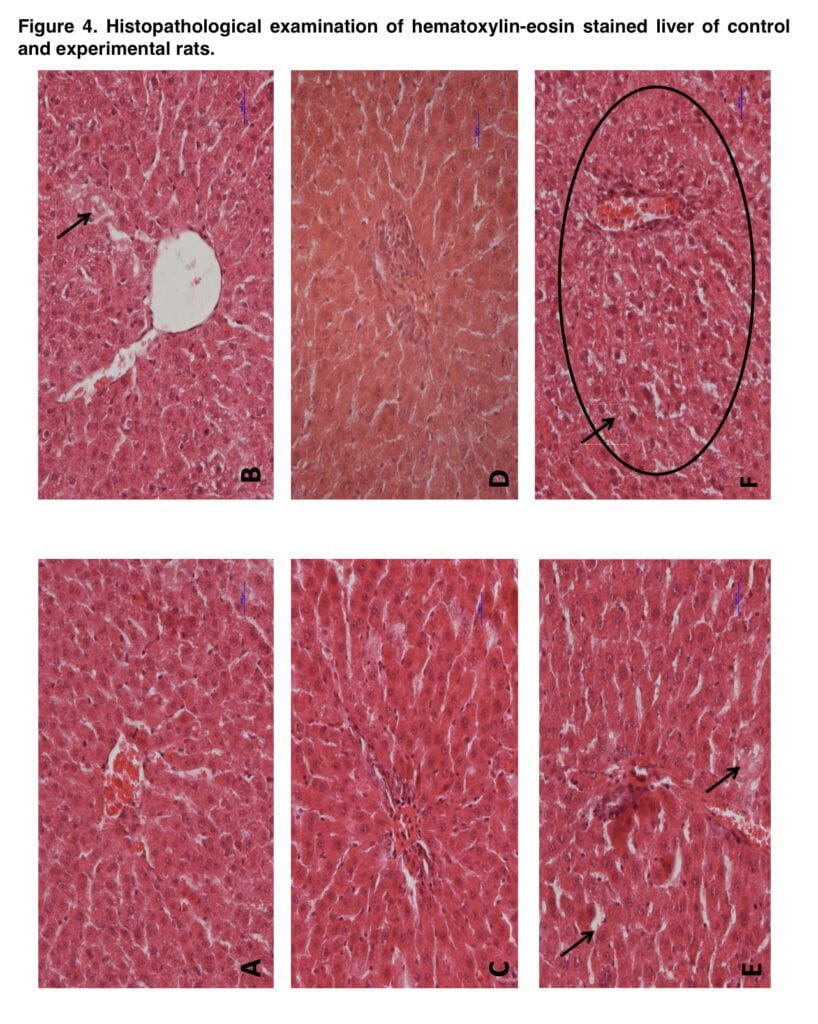
The quantity of DNA collected for analysis was calculated so that the final DNA concentration following dilution in binding solution was 150 ng/μl. The total DNA methylation level was expressed as the relative methylation level in the samples with respect to the methylated control. The taken kidney and liver samples were subjected to histopathological examination. The material was fixed in 4% buffered formalin, dehydrated, and paraffin-embedded. The 4-μm-thick sections were stained with haemotoxylin and eosin for histopathological examination under light microscopy.
The results of our experiment, their analysis and discussion
In the current investigation, behavioral and epigenetic tests were used to examine the effects of chronic mephedrone exposure during adolescence on the rewarding effect of morphine in adult rats. Additionally, a histological investigation looked at the impact of teenage exposure to mephedrone on the architecture of renal and hepatic tissues.
A time of transition between childhood and maturity is adolescence. In rodents, it is classified as PND 28–45, but in humans it is thought to span from 12 to as late as 25 years of age. Important physiological changes in brain functioning take place at this time. This era also corresponds with the peak time for drug use in humans, which may have an adverse effect on brain function and alter drug usage in adulthood. Mephedrone is a stimulant substance that has chemical properties with amphetamine and has effects that are comparable to those of cocaine and amphetamine.
Mephedrone causes short-lived (from 1 to 4 h) psychedelic effects in humans, which are frequently linked to compulsive “re-dosing”. There isn’t a perfect animal model for mephedrone administration that can perfectly replicate human use of the drug. Therefore, in the current investigation, we supplied the effective dosages (10 mg/kg) of mephedrone to adolescent rats over the course of seven consecutive days based on the pharmacokinetic profile of the short-acting medication mephedrone and literature data detailing amphetamine effects in young animals. This method of mephedrone delivery can be compared to the regular exposure of young people to an effective dosage of the drug.
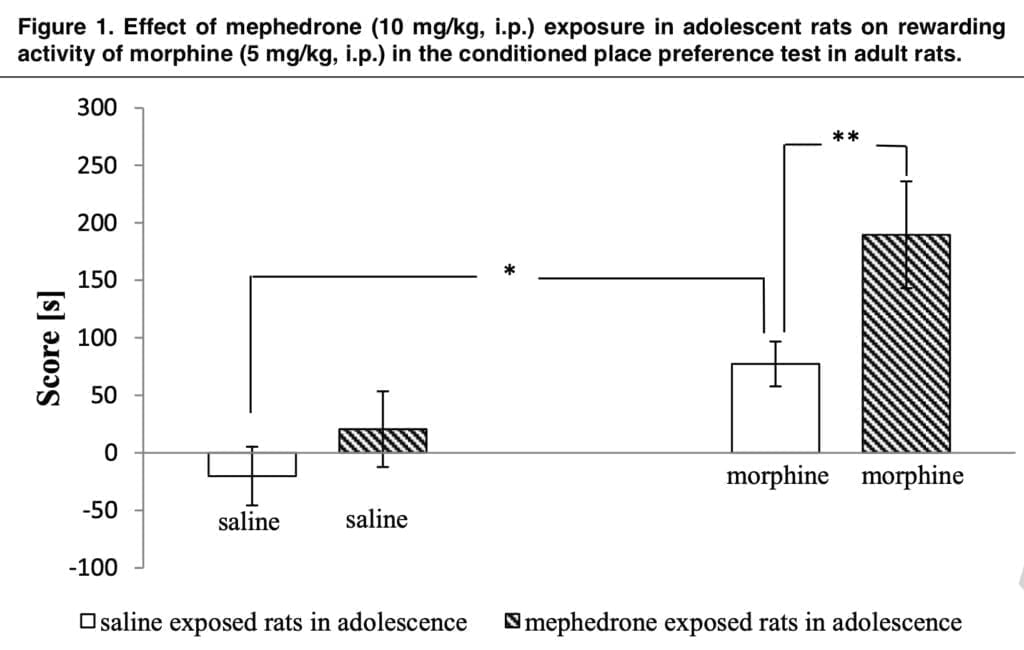
The CPP test was then carried out on the animals once they had reached adulthood. Because the mephedrone-exposed rats spent a lot longer time in the morphine-paired compartment than their saline-exposed counterparts did, we first showed in this test that mephedrone administration beforehand amplified the rewarding impact of morphine in these animals. Notably, the investigated animals’ locomotor activity remained unaffected.
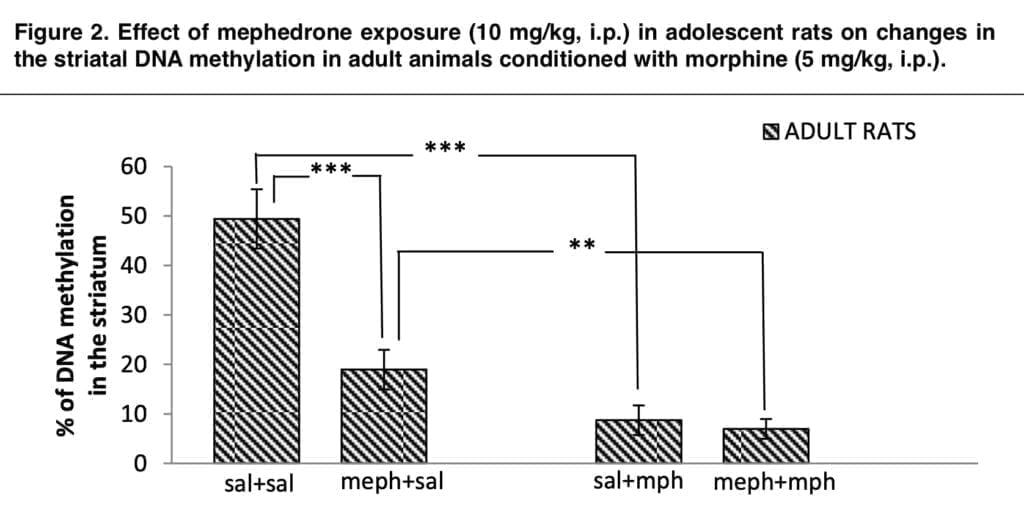
Our findings offer the first concrete proof of the impact of teenage mephedrone exposure on morphine levels. These findings emphasize mephedrone as a gateway drug by indicating that teenage exposure to mephedrone changes responsiveness to the rewarding characteristics of substances that are often misused. We looked at the DNA methylation level in the second portion of our study. Thus, we were able to demonstrate that teenage exposure to mephedrone drastically decreased the overall DNA methylation in the striatum, a part of the brain that is important for addiction.
Adult rats showed this decrease in DNA methylation, while adolescent rats did not. Even while conditioning with morphine in rats that had been exposed to mephedrone for three days lowered the global DNA methylation in a substantial way, conditioning with morphine reduced it more dramatically than conditioning with mephedrone alone. Thus, we showed that adult rats’ DNA methylation was significantly altered by combining teenage mephedrone exposure with morphine training. Different types of gene activity disruption may result from this.

DNA methylation is one of the epigenetic mechanisms which produces heritable changes in the genome. These alterations may affect the phenotype, without effect on DNA sequences. Various environmental influences, including as the use of addictive substances, are known to promote epigenetic processes, which may then regulate gene expression to cause neuroadaptations.
For instance, after the injection of psychostimulants to several components of the dopamine mesolimbic system in adult and adolescent rats, substantial changes in DNA methylation were seen. The amygdala, hippocampus, prefrontal cortex, nucleus accumbens, and striatum are a few examples of these structures. In addition, rats exposed to nicotine and amphetamine for 14 days experienced long-lasting alterations in gene expression and DNA methylation. However, not in the prefrontal cortex, cocaine-induced self-administration in rats also resulted in a decrease in DNA methylation there.
Our work is the first to show the long-lasting changes in DNA methylation in striatum following exposure to mephedrone, as there are currently no data on the impact of mephedrone on DNA methylation in animal tissues. Although the precise processes by which mephedrone reduces DNA methylation in adult rats are unknown based on the results of our investigation, we proposed two routes as potential explanations.
Mephedrone-induced MeCP2 level drop is one option, while a decrease in the activity of DNMT3A, one of the enzymes involved in DNA methylation, is another. It may have an impact on some DNA regulatory or promoter regions and suppress the production of transcription factors, coding enzymes, and enzymes implicated in the rewarding and addictive effects. The confirmation of this hypothesis requires further studies. Then, we looked at the structural changes to the liver and kidney, two significant peripheral organs.
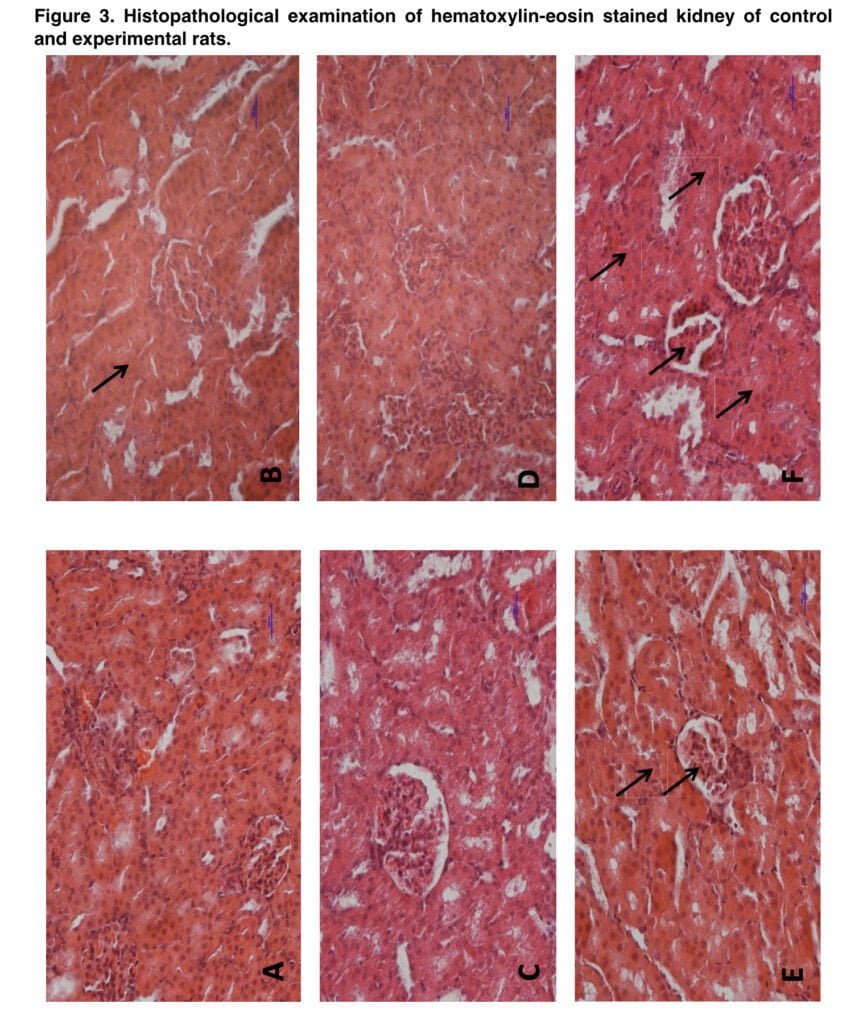
Our study showed for the first time that exposure to mephedrone during adolescence caused local morphological alterations that, according to our theory, were diminished in adult animals due to the tissues’ potential for regeneration. However, certain local retrogressive alterations were seen following the injection of morphine alone, supporting the findings of other writers about the toxicity of morphine. Furthermore, training with morphine in adult rats who had previously received mephedrone treatment resulted in the highest retrogressive abnormalities in the kidney and liver.
This results significantly supported the idea that mephedrone enhanced morphine-induced problems. Thus, we showed that mephedrone exposure during adolescence may result in long-lasting changes in peripheral organs. It is obvious, that the renal and hepatic impairments have no direct effect on the rewarding effect of morphine in our study, however, indirect connections (such as reduced morphine elimination) cannot be excluded.
Conclusion
In conclusion, the mesolimbic system in the brain can be affected by mephedrone, a psychostimulant substance. The following findings back up the idea that mephedrone exposure throughout adolescence is bad for the brain and other organs:
- This impact is proven in the CPP test by the increase of rewarding activity of morphine in adult rats. It may result in long-term sensitization of the neurons to the action of other abused substances (such as morphine).
- It may cause changes in the striatal DNA methylation, which may modify the way that genes are expressed (these alterations were seen to be the strongest in the morphine-conditioned rats that were previously treated with mephedrone).
- It may produce some retrogressive disturbances in kidney and liver.
It must be stressed that all of these changes will encourage the cycle of morphine addiction and relapse, favor organ abnormalities, and raise the chance of adverse repercussions later in life following teenage mephedrone exposure.
