While sex and gender variations in the pharmacological effects of traditional stimulants like cocaine and methamphetamine are extensively documented, sex and gender differences in the effects of synthetic cathinones have received less attention and seem to occur less frequently. Boys in high school and middle school were more likely than girls to use synthetic cathinones, although adult male and female 4-methylmethcathinone use patterns were comparable.
Due to potential negative effects on their health, women were more likely than males to stop taking synthetic cathinones. Studies (Mephedrone Research) on synthetic cathinones conducted on animals reveal ambiguous sex differences. In one study, male and female mice exhibited identical levels of α-PVP-induced locomotor activity, however in a different study, female rats exhibited higher levels of α-PVP-induced locomotor activity than men. While both sexes of rats self-administered α-PVP and 4MMC, male rats displayed larger location preference for α-PVP than female rats. Male rats had stronger cardiovascular effects from MDPV and increased heat caused by α-PVP.
Additionally, rats of both sexes exhibited similar location preferences after MDPV, although females had less conditioned taste avoidance than males. This body of literature reveals how little is known about how sex variations in the effects of synthetic cathinones. The main inflammatory cytokines that control how the innate immune system reacts to shock or injury, such as medication exposure, are interleukin (IL)-1, IL-1, IL-6, and tumor necrosis factor α (TNF-α).
Mephedrone research: description
Human studies in the past have shown that stimulant use modifies plasma levels of peripheral inflammatory cytokines. Many abused drugs, including stimulants, activate nuclear factor-κB (NF-κB) transcription leading to increases in NF-κB-regulated pro-inflammatory cytokines IL-1β, IL-6, TNF-α, and chemokine ligand 2, and the level of inflammation appears to correlate with length of abuse. TNF-α seems to be important in the development of dopaminergic neurotoxicity. Furthermore, levels of these inflammatory markers in the brain can fluctuate depending on whether a person is exposed to stimulants acutely or repeatedly.
There is conflicting evidence about the impact of synthetic cathinones on neuroinflammation. Repeated 4MMC experimenter treatment in certain experiments showed no impact on microgliosis or oxidative stress. While repeated 4MMC or MDPV treatment showed no effect on GFAP in the striatum, repeated 4MMC administration enhanced glial fibrillary acidic protein staining in the hippocampus. Antagonism of the chemokine CXCR4 blunted MDPV-induced locomotor activity and place preference, suggesting that CXCR4 plays a pivotal role in the behavioral and rewarding effects of MDPV.
Despite the fact that this body of research offers strong support for the idea that synthetic cathinones can affect inflammatory levels, all of the previous investigations were carried out on male rats, with the exception of Annekan’s. The goal of this study was to determine the profile of inflammatory cytokines in various brain areas and plasma in order to perhaps identify effects of drug exposure that are particular to those parts of the brain and to determine whether these effects varied depending on gender. Rats self-administered saline or synthetic cathinones for varying amounts of time. Because the onset of drug addiction promotes NF-B transcription, proinflammatory cytokines that are controlled by NF-B were investigated in several brain areas and plasma.
Design of the experiment conducted
In our study, we used adult male and female rats about 65-70 days old. α-Pyrrolidinopentiophenone (α-PVP) and mephedrone (4MMC) were synthesized in house using standard synthetic procedures. They were formulated as recrystalized salt and were > 97% pure. The purity was assessed by several analytical techniques including carbon, hydrogen, nitrogen (CHN) combustion analysis, and proton nuclear magnetic resonance spectroscopy.
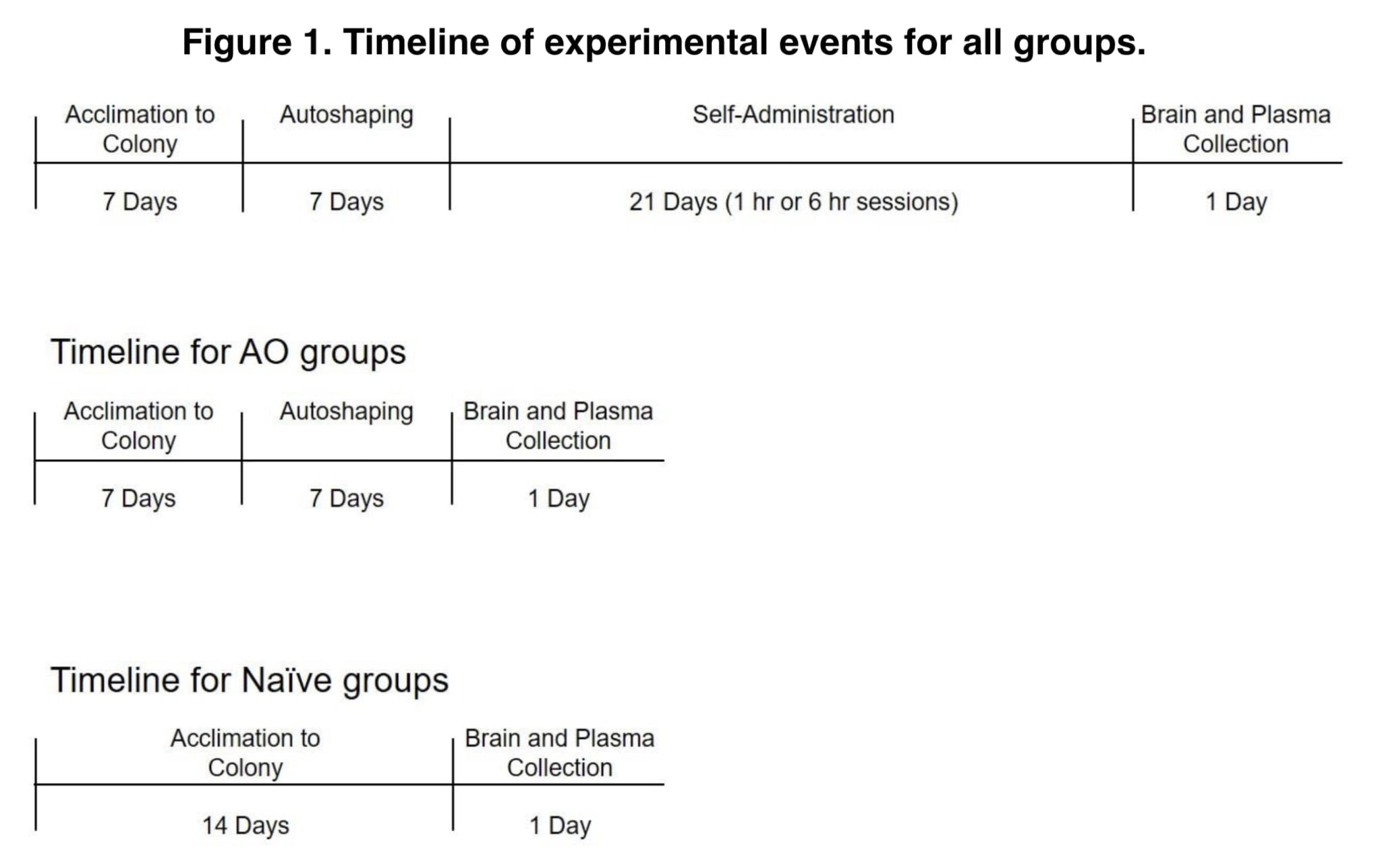
Rats were randomly assigned to one of nine experimental groups: α-PVP autoshaping only (AO), 4MMC AO, saline AO, α-PVP short access, 4MMC ShA, saline ShA, α-PVP long access, 4MMC LgA, or drug- and experimentally-control. All α-PVP groups had access to 0.1 mg/kg/infusion, and all 4MMC groups had access to 0.5 mg/kg/infusion. Through an autoshaping process, rats in all groups were taught to administer themselves for a week.
During autoshaping sessions, an active lever extension and an infusion based on a 60-second random time schedule were combined. An infusion was given fifteen seconds after lever extension or right after following a lever push. A 20-s break period after infusions was indicated by the lighting of both stimulus lights. 15 infusions were administered during autoshaping sessions during a 30-minute period. Sessions ended for rats in all AO groups after autoshaping. For an additional 21 days, the ShA and LgA groups continued to self-administer on a fixed ratio 1 schedule of reinforcement (FR1) during daily sessions lasting either 1 hour or 6 hours.
To prevent the direct effects of the drugs, rats in the self-administration groups were quickly decapitated around 24 hours after their final self-administration session, while rats in the native group were killed after 14 to 15 days in the colony.
On the ventral side of the brain, the hypothalamus was separated into two parts. The hippocampus was then removed after the brain was split along the middle and each cortical half was opened. Tissue samples were wrapped in aluminum foil, inserted into a cryovial, flash frozen in liquid nitrogen, and stored at −80°C. Whole trunk blood was collected and centrifuged for 10 min at 3,000 rpm. Plasma was extracted and stored at −80°C. Secreted protein expression profiling of inflammatory cytokines was performed for plasma and brain tissue.
Inflammatory cytokines in plasma and brain tissue were examined using rat antibody cytokine slide arrays from the RayBiotech G2 series. Arrays were blocked using a 1X blocking buffer at room temperature for 60 minutes on a platform rocker. The array wells were then cleared of blocking buffer. Samples were placed on arrays and incubated at 4°C on a platform rocker throughout the whole night. Arrays were scanned at a wavelength of 532 nm using 2-color fluorescent detection.
Results and discussion
All cytokines that were measured had their protein levels altered by synthetic cathinone exposure. When these changes occurred, they were mostly characterized by increases in cytokine levels compared to the corresponding saline control groups, though some decreases in cytokine levels were also noted. Every measured area of the brain and the plasma saw relative changes in protein levels at some time during the investigation.
Despite a few exceptions, the majority of the changes in cytokine levels for synthetic cathinone groups were seen in LgA but not ShA groups. LgA self-administration was particularly impactful for 4MMC, for which drug effects occurred almost exclusively in LgA groups but not ShA groups. Furthermore, when results were compared to the corresponding saline groups, administration of ShA and LgA to synthetic cathinones resulted in more significant alterations in cytokine levels than exposure to AO. These findings collectively imply that increased exposure to synthetic cathinones is associated with increased levels of neuroinflammatory marker abnormalities.
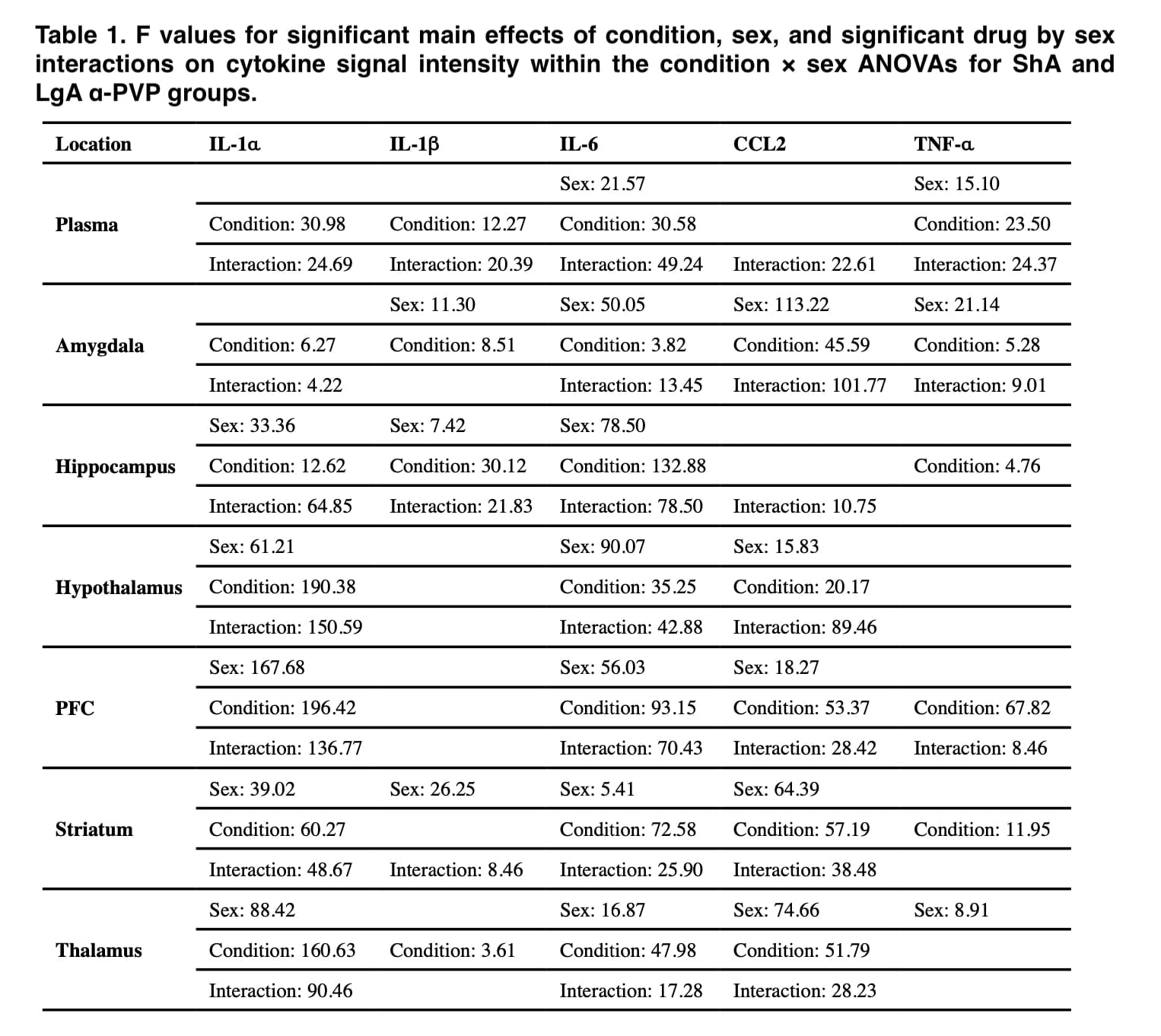
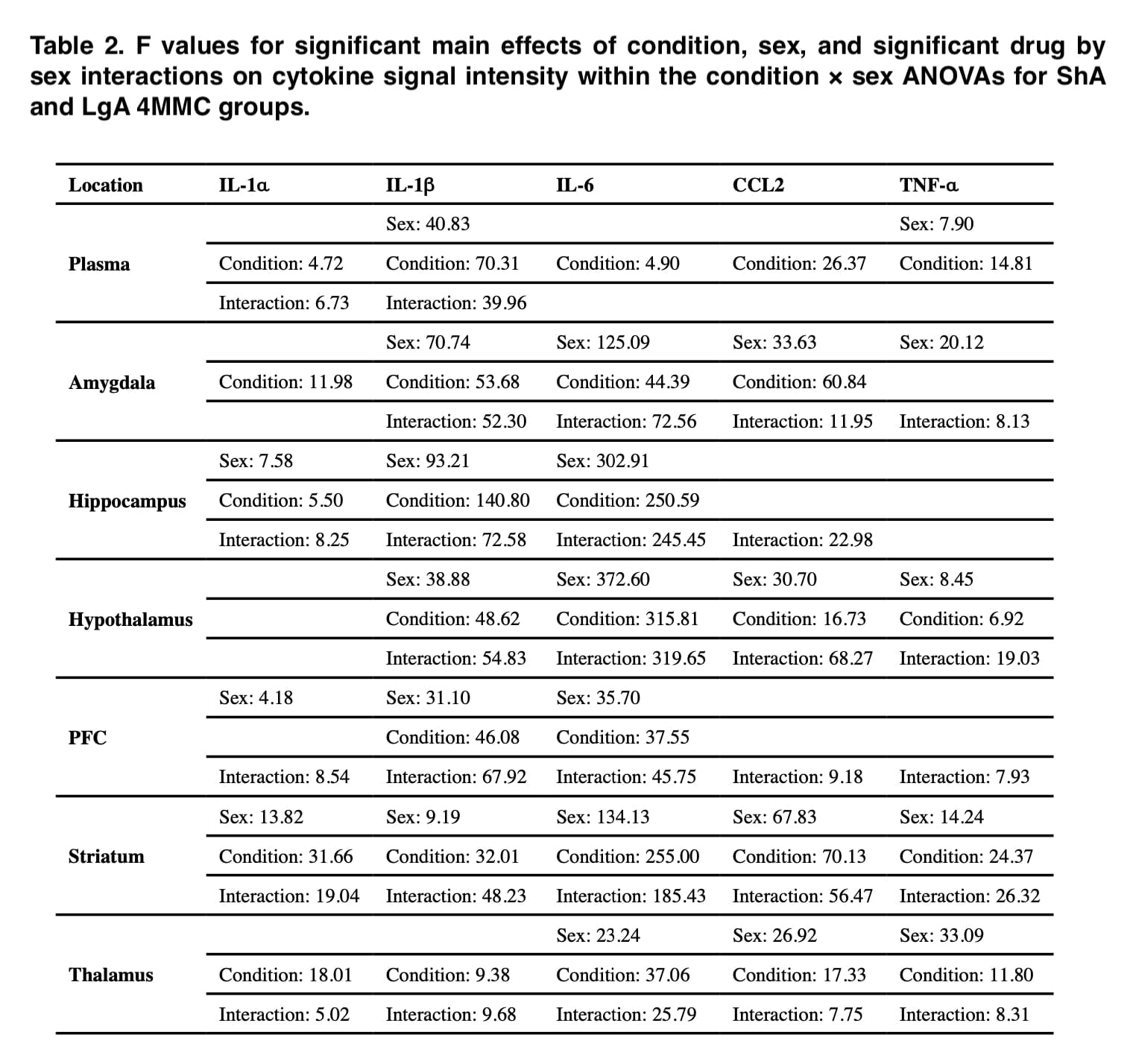
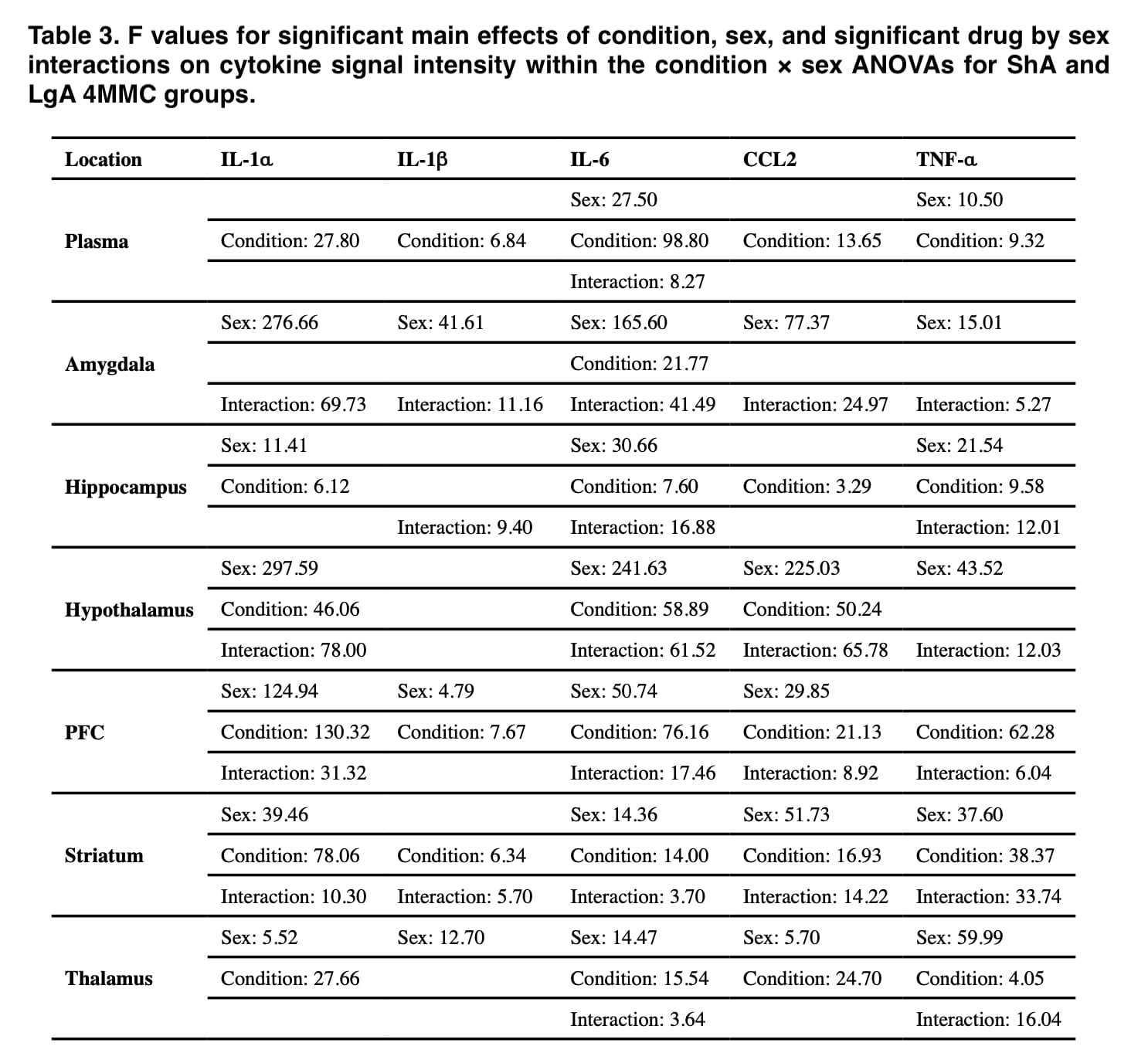
Throughout this investigation, significant cytokine level disparities between the sexes were found. The rats in this study’s self-administration data demonstrate that there were no sex differences in any group’s medication consumption. Contrarily, self-administration of α-PVP and 4-MMC resulted in significant sex variations in IL-1, IL-1, IL-6, CCL2, and TNF-α in the same rats, which cannot be accounted for by sex differences in drug exposure levels.
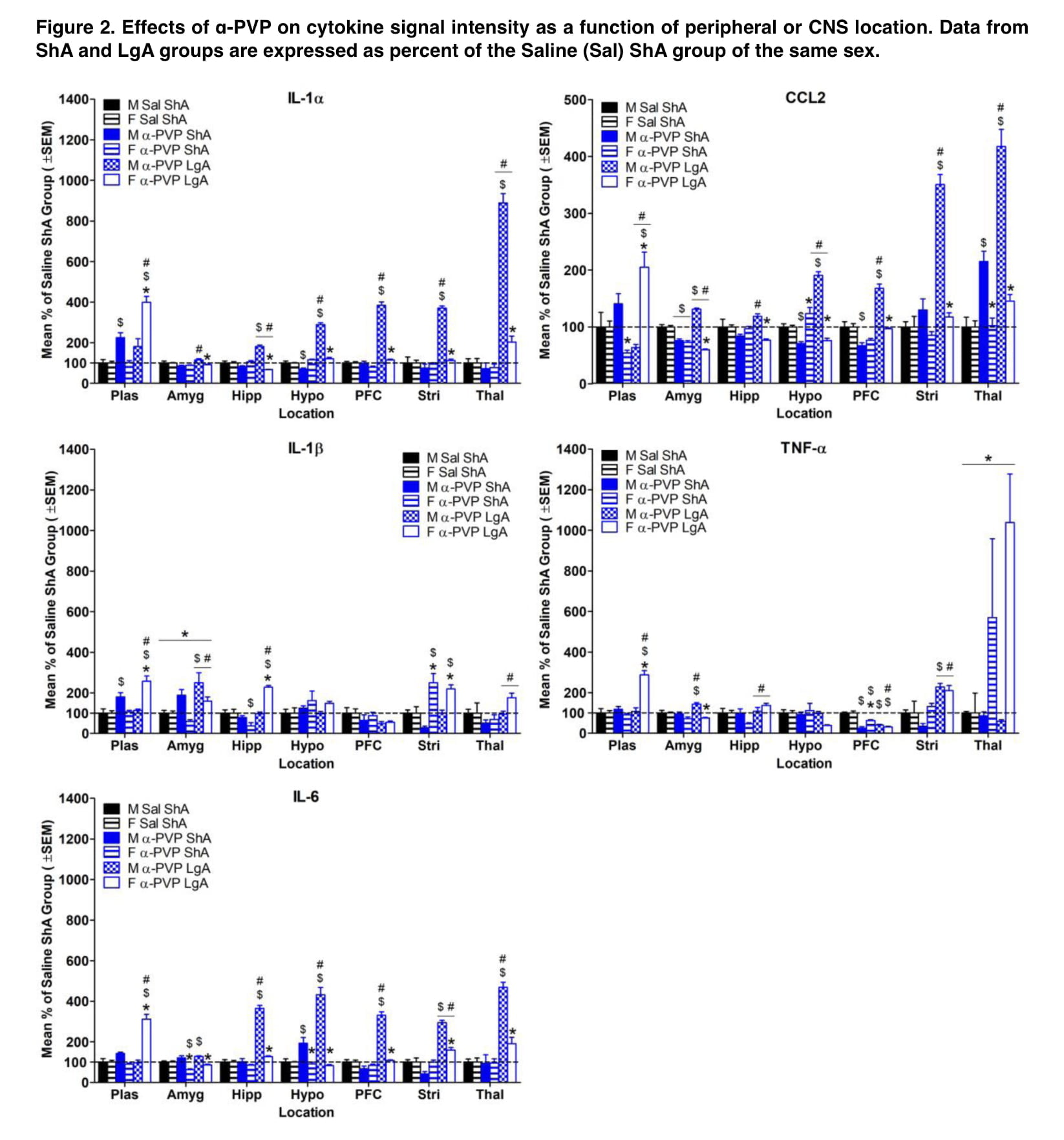
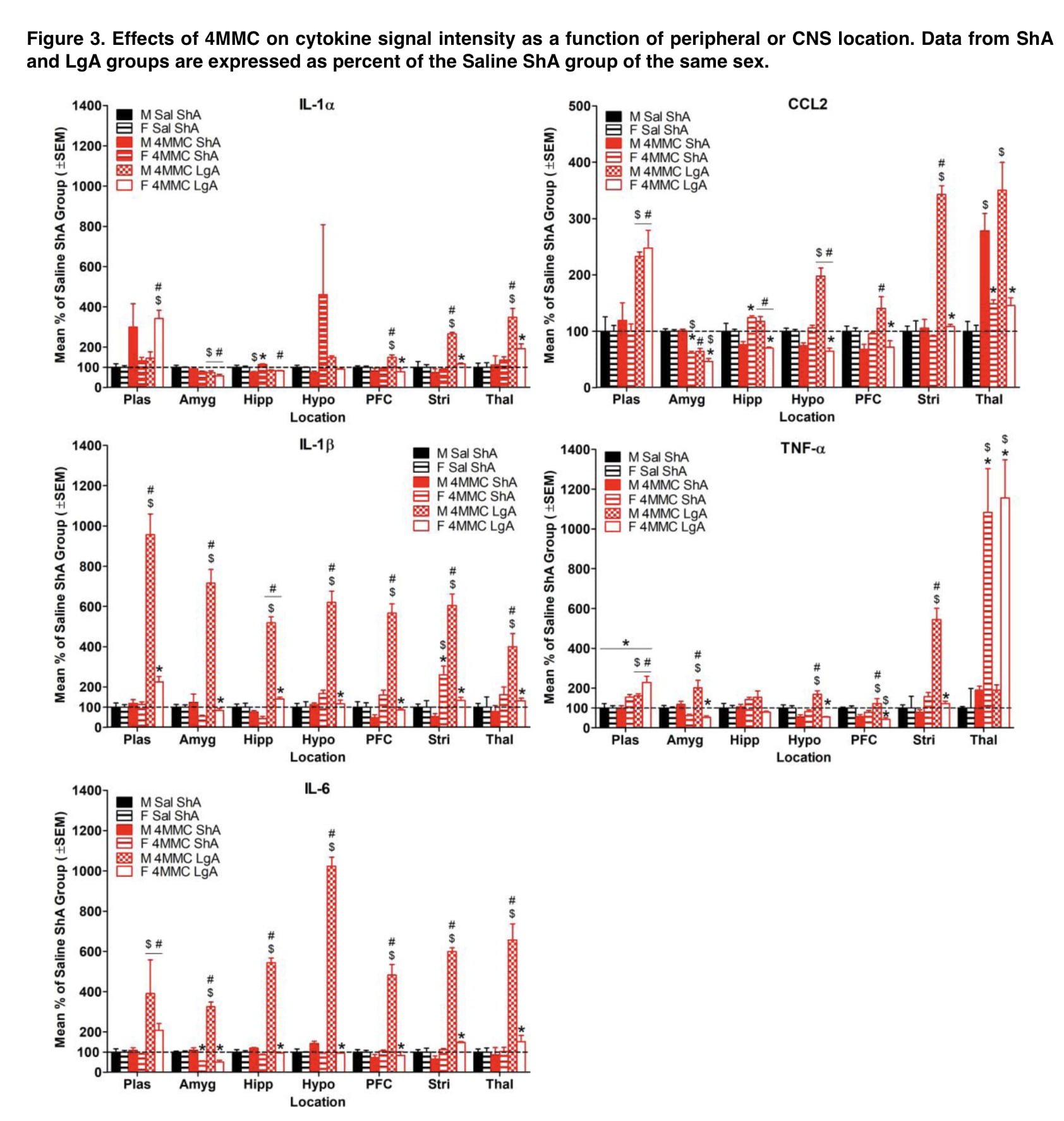
When results were standardized to control groups, male rats had more extensive drug-induced elevations in inflammatory cytokines in their brains than female rats did, whereas female rats were more likely to have these increases in their plasma (Figures 1 and 2). Additionally, these sex disparities in cytokine levels were more frequent following self-administration of LgA synthetic cathinone than ShA, suggesting that sex differences in neuroinflammatory markers may only become apparent after drug consumption has been dysregulated.
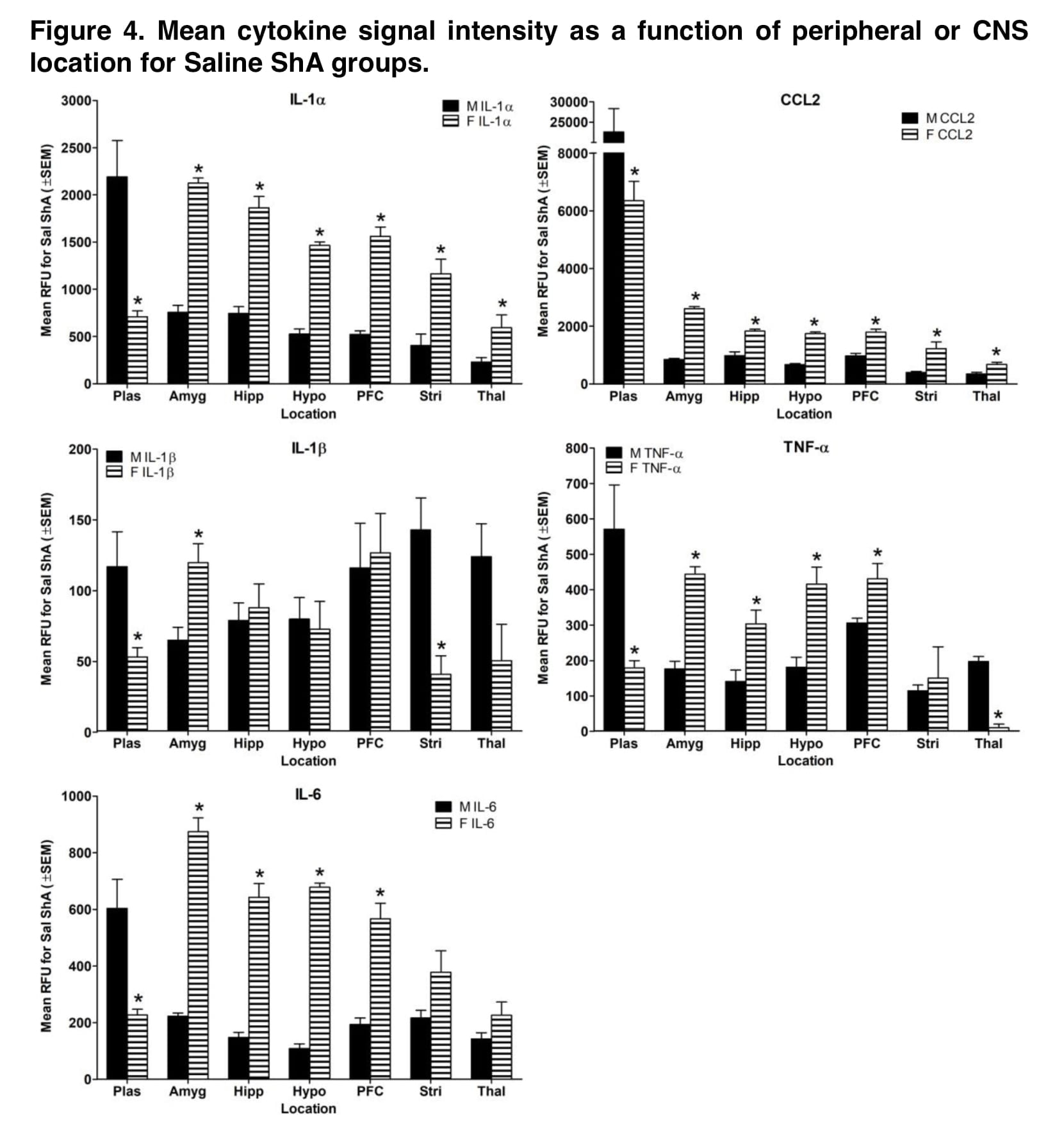
The native groups occasionally saw sex variations in cytokine levels, whereas saline groups saw these differences more frequently (Figures 3). This implies that even in the absence of drug exposure, exposure to experimental methods may result in sex variations in the levels of inflammatory markers.
In addition to sex, the drug’s mechanism of action might be a key element in the development of stimulant-induced neuroinflammation. Different inflammatory profiles were produced by self-administration of the dopamine releaser mephedrone and the dopamine uptake inhibitor α-PVP (Figures 1 and 2). Compared to α-PVP, mephedrone increased cytokine levels by a higher magnitude, and sex differences in cytokine levels were more pervasive across all brain areas. Additionally, IL-1 levels in plasma, the hippocampus, and the striatum were higher in the female α-PVP LgA group than in the male mephedrone LgA group compared to their respective Saline ShA groups.
This shows that the mode of action may contribute to the emergence of neuroinflammation that is particular to sex. The fact that exposure to synthetic cathinone occasionally reduced proinflammatory cytokine levels is another potentially important result of the current investigation. Additionally, it has been shown that proinflammatory markers are downregulated in abstinent cocaine users, which is assumed to be the consequence of compensatory mechanisms that were activated after cocaine usage stopped. Interestingly, decreases in cytokines in the present study were more common during initial drug exposure than following self-administration (Figures 1 and 2). Thus, duration of drug exposure may impact the processes involved in downregulation of inflammatory cytokines.
Because the timepoint at which brain and blood samples were taken differed among trials, comparisons of the current results with earlier cocaine and methamphetamine self-administration research should be made with care. Previous research has looked at inflammatory markers in samples taken within two hours after the last drug delivery, or when animals were really experiencing the effects of stimulants. The present study collected biological samples approximately 24 hrs after the last drug administration in accordance with some previous studies. It is uncertain if the rats in the current study were experiencing withdrawal when brain tissue was taken since it is unknown if synthetic cathinone self-administration causes withdrawal and because withdrawal takes time to develop.
The levels of inflammatory markers were examined in other trials two or more days following the final drug exposure, when drug withdrawal was more likely to be taking place. In the present study, saline groups also showed changes in cytokine levels compared to the native group. Interestingly, saline groups were more likely to show dysregulation in cytokine levels compared to the native groups, unlike the effect in drug groups relative to saline groups.
The fact that the present study failed to pinpoint a particular explanation for the variations in cytokine levels between the saline groups and the native group is one of its limitations. Future research is required to ascertain whether the differences between saline groups and native groups were due to learning—since even saline rats learned to press the active lever to turn on the stimulus lights—or if the implantation of jugular catheters or the chemicals used to keep catheter patency were to blame.
Conclusion
In conclusion, this study demonstrates that self-administration of synthetic cathinones induced significant sex differences in proinflammatory cytokines (IL-1, IL-1, IL-6, CCL2, and TNF-α), which are associated to stimulant use problems. In contrast to female rats, who were more likely to exhibit elevated inflammatory cytokines in plasma as compared to saline groups, male rats had general increases in measured cytokine levels in their brains, notably for mephedrone.
Additionally, LgA had access to synthetic cathinones more often than ShA, demonstrating that sex differences in neuroinflammation may only become apparent after dysregulated drug use. The changes in proinflammatory cytokine levels seen in the current study imply that usage of synthetic cathinones throughout the transition from use (ShA) to abuse likely results in sex-specific patterns of neuroinflammation (LgA). As a result, the necessity for therapy may vary according on sex and the course of synthetic cathinone usage.