When using mephedrone, people often find that short-term memory begins to perform very poorly. What you were told a few seconds ago is easily forgotten. If you read this publication, you will learn why mephedrone reduce memory and how to medicate your brain to help your memory failures.
Predominantly during adolescence, many neurobiological developmental stages can be affected by various environmental factors, including drugs. Current evidence suggests that learning and memory are extremely vulnerable to substance use during adolescence, and similar cognitive impairments may persist into adulthood. The neuroanatomical contributors of learning and memory are abundant, as cognition is a multisystem behavior. Herein, the prefrontal cortex and hippocampus predominate.
The effects of systematic drug use on each subregion of these brain areas contribute to the disruption of their typical development, which will eventually manifest itself as persistent cognitive impairment.

What is memory?
Memory is the sum total of what we remember and gives us the capability to learn and adapt from previous experiences as well as to build relationships. It is the ability to remember past experiences, and the power or process of recalling to mind previously learned facts, experiences, impressions, skills, and habits. It is the store of things learned and retained from our activity or experience, as evidenced by modification of structure or behaviour, or by recall and recognition.
In this sense, it’s clear that memory is not just one thing and memory operates in a complex way. Memory entails autobiographical memory, which is information about ourselves in relation to the world, there’s learning and remembering facts, and even muscle memory that helps us play musical instruments or ride a bicycle.
Much of this enters our long term memory or makes up procedural memory, episodic memory, sensory memory, and explicit memory, to name some. And encoding and retrieval of memory become problematic when it comes to issues of false memories, memory loss, or memory disorders.

Mephedrone Reduce Memory?
Mephedrone use has been shown to be associated with impaired short-term and verbal memory in humans. In rodents, mephedrone causes acute deficits in working memory and impaired object recognition, and selectively impairs fear-motivated contextual memory retention. Such cognitive impairment is often accompanied by changes in striatal quantitative protein levels that are involved in monoamine transport (DAT and SERT).
Matrix metalloproteinases (MMPs) are the main enzymes involved in the reorganization of the central nervous system; they are a family of proteases that degrade components of the extracellular matrix. The best known and most studied are MMP-2 and MMP-9. MMP-9 (gelatinase B) is a zinc-dependent endopeptidase that is present in the hippocampus, cerebellum and cortex and is expressed by neurons and glia
MMP-9 is fundamental to brain development because of its association with neurophysiological functions such as synaptic plasticity and long-term potentiation. In the adult mammalian brain, levels of this proteinase are low, but MMP-9 expression is elevated and enhanced during learning or synaptic potentiation.
Dysregulation of MMP-9 leads to a variety of pathologies including schizophrenia, epilepsy, fragile X syndrome, Alzheimer’s disease, and depression, and abnormal activity of this proteinase contributes to cognitive dysfunction. Published data indicate that mephedrone abuse alters MMP-9 levels in various brain structures.
In this study we used the Barnes maze to assess and study spatial learning and memory in rodents, and examined two hypotheses: whether mephedrone administered during adolescence causes memory impairment in adult rats; and whether the effects of mephedrone are similar to those of amphetamine. In addition, we performed a series of neurochemical experiments to examine mephedrone and amphetamine levels on MMP-9 levels (with ELISA test) as well as expression of postsynaptic density protein-95 (PSD-95) in the hippocampus in rats.

Methods Of Research
Healthy Wistar rats were used in this study. Mephedrone hydrochloride and amphetamine hydrochloride were dissolved in sterile isotonic saline and prepared ex tempore before each injection. The dose for mephedrone and amphetamine was 30 mg/kg and 2.5 mg/kg, respectively, and was administered intraperitoneally. The following etological systems and methods were used for the tests:
- The Barnes Labyrinth (with the implementation of the habituation and acquisition phases, as well as the reverse-learning phase);
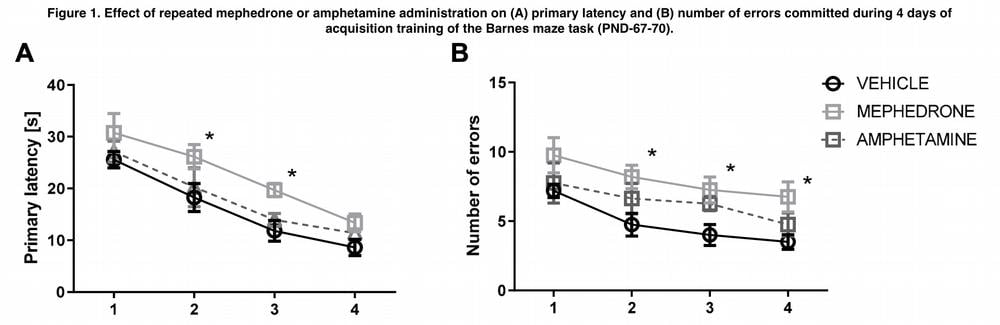
- Locomotor activity identification test;
- Western Blot (the tissue was homogenized brain structures);
- ELISA Assay (quantification of MMP-9 in tissue homogenates).
Results and discussion
In our study we saw that rats exposed to mephedrone and amphetamine in late adolescence show deficits in spatial learning in adulthood. Mephedrone administration causes changes in MMP-9 in the hippocampus and prefrontal cortex, two areas that are crucial in the formation and recovery of long-term spatial memory.
In adult individuals (PND87) with impaired memory, significant changes were observed in the expression of NMDAR subunits, as well as in the expression of PSD-95. Overall, the results suggest that recreational use of mephedrone during adolescence may cause cognitive impairment of spatial memory, which will manifest in later adulthood.

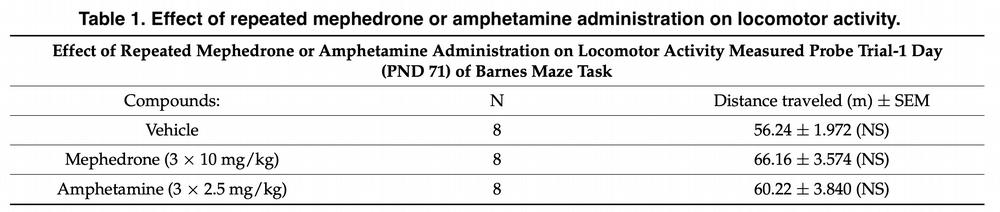
In our experiment 1, adult rats that were repeatedly exposed to mephedrone during adolescence (PND 40-49) were found to show significant deficits in acquiring “new” spatial memory, and there was an increase in primary latency and error rates during a trial in the Barnes maze system. Similar phenomena were also observed during prolonged withdrawal (for three and five weeks – Trial 2).
Thus, given that the above-mentioned memory disturbances were long-lasting, we can conclude that such cognitive deficits are the result of neurocognitive dysfunction due to neuronal changes. We have shown for the first time that mephedrone, unlike amphetamine, has a much more deleterious effect on memory, especially when used in adolescence.
It has been observed in recent publications that mice given mephedrone at a dose of 30 mg/kg eight weeks prior to the experiment showed controversial results, such as improved memory during a reversal learning test in MWM. This is likely due to the fact that mephedrone was given to adult mice. Therefore, this result may be due to the animals’ ability to learn a new location. In our study, mephedrone was administered to those adolescent rats who were still developing brain structures.
Rats injected with more mephedrone than amphetamine had difficulty correcting their response when changing the position of the escape box in the reverse learning test. This was because the animals developed severe deficits in memory processes associated with learning information on the day of testing (Trial 1 and 2).
It is known that MMP-9 proteinase activity in brain tissues is significantly increased during hippocampus-dependent memory tasks, especially after excitotoxic exposure. As shown in our tables, in experiment 2, MMP-9 levels increase in the hippocampus and prefrontal cortex after 2-5 weeks of withdrawal of mephedrone administration.

The prefrontal cortex appeared to be the more sensitive part of the brain, as an increase in MMP-9 was observed as early as 24 hours after the last injection of mephedrone. Thus, the changes in MMP-9 levels were unrelated to animal behavior and were probably the result of mephedrone-induced cognitive impairment. As for the rats injected with amphetamine, no changes in MMP-9 levels were observed at the dose used. Therefore, MMP-9 cannot be used as a marker of neurodegeneration in amphetamine use.
If we disassemble the process at the level of secondary messengers, after the release of MMP-9 from the postsynaptic section of excitatory synapses, its activation occurs through the cleavage of specific target proteins (an integrin ß1-dependent pathway). This pathway regulates NMDAR mobility and function in the synapse and is a powerful regulator of the surface movement of these receptors.
However, overexpression of MMP-9, on the contrary, leads to an increase in basal excitatory synaptic transmission and impairs synaptic plasticity. In our study, withdrawal of mephedrone injection caused an increase of MMP-9 level already after 24 hours in the hippocampus and after 14 days in the prefrontal cortex, which actually worsened the cognitive processes in the experimental individuals.
Excessive activity of this marker can lead to morphological and functional changes in the synapses of the studied brain areas. Notably, mephedrone and amphetamine have unique, age-dependent effects on glutamate in these brain structures. Therefore, mephedrone use coexists with a large release of both glutamate and MMP-9 in the hippocampus and prefrontal cortex.

In experiment 1, the GluN2B subunit in the hippocampus and prefrontal cortex was increased in adult rats treated with mephedrone, whereas the GluN2A subunit in the hippocampus was decreased in those rats treated with amphetamine. Our published data indicate that expression of the GluN2B subunit in the synaptic membrane decreases during aging, and increased association of GluN2B-containing NMDAR with PSD-95 in older animals may have contributed to the development of spatial memory impairment.
Our current study demonstrates an increased PSD-95 and GluN2B expression in the PC of adult rats. Such outcome can suggest the presence of this receptor subunit (in the synaptic membrane) and decreased process memory. Hence, these data support our hypothesis that deficits observed in the Barnes maze task (Experiment 1) in the reversal learning (that is cortex-dependent process) may have been due to an impairment of spatial memory processes rather than cognitive flexibility impairment.
In our hippocampal study, the GluN2B subunit expression was increased, but PSD-95 expression was decreased during the mephedrone withdrawal. Such data suggests that the GluN2B subunit is located mainly outside the synapses and might be a consequence of excessive glutamate release in this region. The observed effect seems to be related to the learning and memory impairment observed in Probe trials 1/2.

Conclusion
In conclusion, our study shows that repeated administration of mephedrone, and to a lesser extent, amphetamine, during adolescence, causes spatial memory deficits in adult rats. Impaired cognitive plasticity is a consequence of impaired and impossible spatial learning. In addition, mephedrone use alone resulted in a delayed increase in MMP-9 in the prefrontal cortex and hippocampus.
In contrast, adult rats tested with the Barnes maze and injected with mephedrone showed memory impairment accompanied by increased levels of MMP-9 and changes in the PSD-95-expressing NMDAR subunits. Thus, we can clearly state that the use of mephedrone during adolescence has an extremely negative effect on neurocognitive processes later in life, and such changes are associated with hyperexpression of MMP-9 and dysregulation of the GluN2B subunit in the prefrontal cortex and hippocampus.
